Multiple Sclerosis
Using Reprogrammed Stem Cells as a Therapy for MS



Posted March 8, 2019
Stefano Pluchino, M.D., Ph.D., University of Cambridge, United Kingdom
Frank Edenhofer, Ph.D., Julius-Maximilian University of Würzburg, Germany
Regina Armstrong, Ph.D., Uniformed Services University of the Health Sciences, United States



Multiple Sclerosis (MS) is the most common cause of chronic neurological disability of early-to-middle adult life. The disease process is driven by both persistent inflammation and a concurrent inability of cells within the central nervous system (CNS) to replace damaged myelin (remyelination). Currently, there is no cure for MS, and disease progression is irreversible. Recent advances in stem cell-based therapies led Dr. Pluchino to explore the use of neural stem cells (NSCs) as a potential approach to treating MS. NSCs are “master cells” that can develop into new neurons and myelin-forming cells known as oligodendrocytes.
Dr. Pluchino previously harvested NSCs from a stem cell-enriched region of the adult healthy brain, expanded these in the laboratory, and transplanted them into animal models of CNS disorders, including MS. These studies showed that transplantation of adult NSCs in experimental autoimmune encephalomyelitis (EAE) mice, an animal model of MS, reduced functional impairments, as well as promoted remyelination and recovery of demyelinated neurons. Unfortunately, the use of NSC therapies in humans is limited by several factors: (1) the necessity of human NSCs to be derived from embryos or fetuses and consequent ethical concerns, (2) the difficulty of growing large numbers in laboratories, and (3) the potential to cause immune rejection.
With support from a Fiscal Year 2014 Investigator-Initiated Partnership Award, Dr. Pluchino, along with his partners Dr. Edenhofer and Dr. Armstrong, aimed to identify the ideal source of stem cells for transplantation and to further define their mechanisms of action in the context of MS pathophysiology. First, the team established methods to induce both mouse and human skin fibroblasts to become neural stem cells (iNSCs). When iNSCs were transplanted in the brains of EAE mice through cerebrospinal fluid (CSF) circulation, they produced the same benefits as the NSCs. There was a significant reduction in clinical deficits, as well as spinal cord demyelination and axon loss. Additional ongoing work investigated the effects of iNSC transplantation on functional recovery in a second MS animal model of demyelination. The results showed that iNSC transplantation improved coordination, with some improvements also evident in brain imaging of these mice.
More importantly, these recovery effects occurred concomitantly with reduction of succinate in the CSF of the iNSC-transplanted mice. Since succinate has been linked to metabolic distress and inflammatory activity in MS, the team looked into the mechanism that led to that reduction. They were able to show that transplanted iNSCs can sense the excess of succinate in the inflamed CNS and reduce its levels by initiating an anti-inflammatory effect that begins with binding succinate to the succinate receptor, (SUCNR1)/GPR91, on iNSCs. In turn, this causes iNSCs to release the anti-inflammatory prostaglandin, E2. Notably, succinate may serve as a biomarker of brain inflammation; thus its reduced levels in the CSF may be associated with improvement of MS-associated clinical deficits and neuropathological features.
Human iNSCs are currently being tested in laboratory animals with MS-like lesions, and iNSC therapeutics are under development to be tested in humans. The above studies provide proof-of-concept for utilization of innovative stem cell technologies in experimental MS. These findings from the collaborative work of Drs. Pluchino, Edenhofer, and Armstrong hold promise for translating state-of-the-art cell reprogramming technologies into next-generation, patient-specific, human iNSC therapeutics for progressive MS and other chronic degenerative neurological diseases.
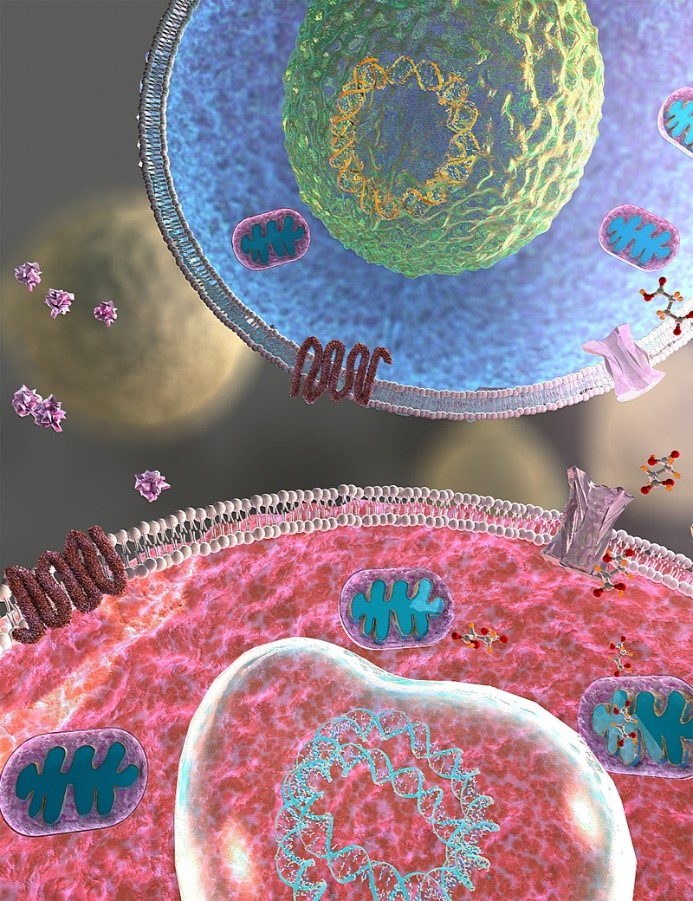
3D rendering of the SUCNR1-succinate axis between grafted somatic or directly-induced NSCs (top) and endogenous inflammatory macrophages/microglia (down). Succinate released by endogenous inflammatory macrophages/microglia activates SUCNR1/GPR91 on NSCs to initiate the secretion of PGE2 and the scavenging of extracellular succinate.
References:
Peruzzotti-Jametti L, Bernstock JD, Vicario N, et al. 2018. Macrophage-derived extracellular succinate licenses neural stem cells to suppress chronic neuroinflammation. Cell Stem Cell 22:355-368.e13. PMID: 29478844.
Peruzzotti-Jametti L and Pluchino S. 2018. Targeting mitochondrial metabolism in neuroinflammation: towards a therapy for progressive multiple sclerosis? Trends Mol Med 24(10):838-855. PMID: 30100517.
Peruzzotti-Jametti L, Bernstock J, Vicario N, et al. 2017. Transplanted induced neural stem cells ameliorate experimental autoimmune encephalomyelitis by metabolic reprogramming of mononuclear phagocytes. GLIA 65:E401-E402.
Vicario N, Peruzzotti-Jametti L, Braga A, et al. 2017. Transplantation of directly induced neural stem cell (iNSCs) promotes remyelination in a mouse model of experimental focal demyelination. GLIA 65:E400.
Volpe G, Bernstock JD, Peruzzotti-Jametti L, and Pluchino S. 2016. Modulation of host immune responses following non-hematopoietic stem cell transplants: translational implications in progressive multiple sclerosis. J Neuroimmunol S0165-5728(16)30312-5. PMID: 28034466.
Link:
Last updated Monday, March 10, 2025














