Multiple Sclerosis
Link Between Alterations in the Gut Microbiota and Multiple Sclerosis



Posted February 28, 2018
Kouichi Ito, Ph.D., Rutgers-Robert Wood Johnson Medical School

Multiple sclerosis (MS), an autoimmune disease that targets the central nervous system (CNS) and causes inflammatory demyelination and axonal loss, commonly affects young adults. Symptoms vary from person to person and include fatigue, weakness, numbness or tingling, and difficulty walking. Environmental and genetic factors, as well as age and microbial infections, have all been implicated in the development of autoimmune diseases. Recent studies also suggest that dysbiosis, or alterations in the gut microbiota, influence the onset and progression of autoimmune diseases, including MS. Although it is recognized that dysbiosis promotes a proinflammatory immune cell phenotype, the exact mechanism(s) involved in the initiation and progression of autoimmune diseases are unknown.
With support from a Fiscal Year 2011 Idea Award, Dr. Kouichi Ito and his team used an animal model of MS to investigate the effects of gut dysbiosis and age on the development of CNS autoimmunity. In a recent Proceedings of the National Academy of Sciences of the United States of America (PNAS) publication, fecal samples analyzed from transgenic mice expressing HLA-DR2a and T cell receptors specific for myelin peptides isolated from MS patients showed that gut dysbiosis caused by the overgrowth of the bacterial species, Bacteroides vulgatus, during adolescence and young adulthood resulted in the development of spontaneous experimental autoimmune encephalomyelitis (EAE), an animal model of MS (Figure 1). Dr. Ito and his team further elucidated that EAE results from the decreased expression of Foxp3, CBLB, and ITCH genes in T cells, which alters the immunological tolerance to myelin basic protein. Additionally, increased intestinal permeability and blood endotoxin levels were associated with the development of spontaneous EAE, supporting the hypothesis that gut dysbiosis is one of the risk factors associated with the onset of CNS autoimmunity.
The findings from Dr. Ito and his team highlight the importance of the gut microbiota in autoimmune disease progression. In addition, these findings suggest that gut dysbiosis may play a pathological role in the initiation and progression of MS during adolescence and young adulthood. Dr. Ito has obtained an R21 grant from the National Institute of Allergy and Infectious Diseases to further investigate the link between gut dysbiosis and the pathogenesis of MS. He plans to determine whether gut dysbiosis precedes MS or MS leads to gut dysbiosis. Dr. Ito and his team also plan to investigate whether modifying the intestinal microbiota via diet can reduce the risk of MS.
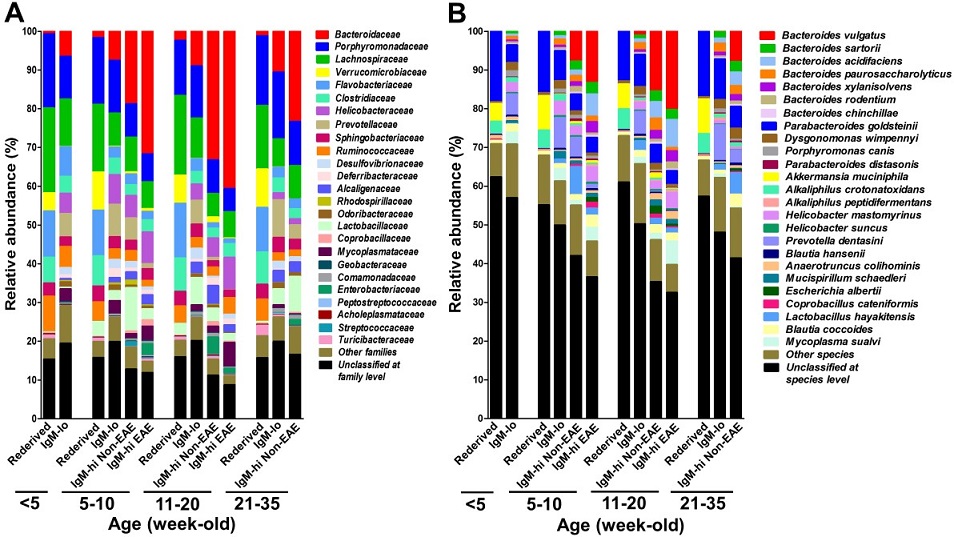
Figure 1. Identification of enteric bacteria expanding in spontaneous EAE mice.
Gut dysbiosis was examined by measuring the levels of fecal IgM every week, and fecal DNA was isolated from re-derived, IgM-lo, IgM-hi non-EAE, and IgM-hi EAE 3A6/DR2a Tg mice at different ages. Enteric bacterial families (A) and species (B) were identified by Illumina 16S rRNA sequence analysis. Gut dysbiosis caused by expansion of the pathogenic enteric bacteria, Bacteroides vulgatus, was highly associated with development of spontaneous EAE.
Reference:
Yadava SK, Boppanaa S, Itoa N, et al. 2017. Gut dysbiosis breaks immunological tolerance toward the central nervous system during young adulthood. PNAS 114(44):E9318-E9327.
Link:
Last updated Monday, March 10, 2025














