Melanoma



Posted April 27, 2022
Sharad S. Singhal, Ph.D., City of Hope National Medical Center, Beckman Research Institute, Duarte, CA
 Dr. Sharad S. Singhal
Dr. Sharad S. Singhal
The first immunotherapy treatment was approved for use in melanoma in 2011, which dramatically improved melanoma patient outcomes. However, about half of melanoma patients with metastatic disease do not respond to immunotherapy.1 Furthermore, melanoma survival rates are highly dependent on the stage at which the cancer is diagnosed. The five-year survival rate for patients diagnosed with localized melanoma is 98%, whereas there is only a 22% survival rate for patients diagnosed with metastatic melanoma.2 These numbers support why, with its inception as a program, the Melanoma Research Program (MRP) issued a Challenge Statement to the melanoma research community to expand the concept of prevention, to include improved detection and monitoring capabilities, as well as inhibiting the initiation of melanoma, the emergence from tumor dormancy, and the development of metastases.
In response to this Challenge, Dr. Sharad Singhal received a fiscal year 2019 Concept Award to elucidate the mechanism of how the oncoprotein ral interacting protein (RLIP) promotes melanoma progression and determine whether RLIP inhibition prevents the development of melanoma. RLIP is an anti-apoptotic (prevents cancer cell death) membrane transporter that serves a critical role in regulating cellular oxidative stress, and is over-expressed in several types of cancers, including melanoma. In the first phase of this project, Dr. Singhal investigated the loss of RLIP using either an antibody that blocked the transporter function of RLIP (anti-RLIP IgG) or a short DNA fragment (RLIP antisense) that prevented expression of RLIP in cancer cells. He used both cell-based (in vitro) and mouse (in vivo) models that also lacked the protein p53, since the loss of p53 is frequently observed in cancer and is associated with cancer progression and metastasis. The results of these studies show that RLIP appears to be necessary for melanoma survival, and that its depletion is sufficient to prevent the spontaneous development of melanoma. These findings represent a novel means of overcoming the loss of p53, which is a barrier to treating many cancers, including melanoma.
Dr. Singhal also studied the depletion of RLIP as a novel therapeutic target to follow-up on previous studies that demonstrated reduction of RLIP sensitizes malignant cells to treatment by inducing apoptosis. In Dr. Singhal's laboratory, two different melanoma mouse models treated with anti-RLIP IgG or RLIP antisense showed significant tumor regression. Additionally, these preliminary studies do not reveal any observed toxicity in response to RLIP depletion. This is a key finding because many other transporter proteins have been considered "undruggable” because of treatment-related toxicities that affect normal cells in addition to cancer cells. Current efforts in Dr. Singhal's laboratory include pursuing additional studies to validate RLIP as a viable therapeutic target and developing possible treatment strategies.
In summary, these exciting findings lay the foundation for addressing two critical needs for the melanoma community.3, 4 First, the observation that RLIP inhibition can prevent melanoma formation could be further developed into much needed therapeutic prevention agents. And second, Dr. Singhal's treatment of melanoma tumors with RLIP paves the way for a potentially novel treatment option for patients with metastatic melanoma who do not respond to immunotherapy.
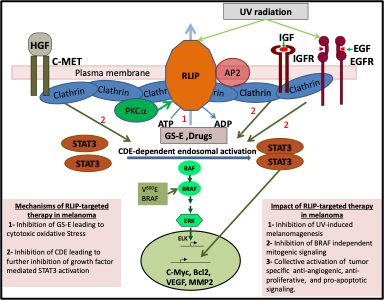
Figure 1: Graphical abstract (provided by Dr. Singhal): Novel mechanisms of action of RLIP-targeted therapy in melanoma. The clathrin-dependent endocytosis (CDE) -mediated activation of mitogenic signaling, induced by a combination of oxidative stress and growth factor signaling, represents a novel and critical axis of tumor signaling that can be targeted to develop effective therapeutic interventions for aggressive melanomas. Current therapeutic strategies focus on inhibiting single components of the melanoma-specific signaling pathways. The high expression of RLIP and the regulation of CDE by its glutathione-electrophile conjugate (GS-E) transport function, as evidenced by our recent studies, have unveiled a unique and potent anti-tumor target in melanoma. A single drug like R508 (RLIP antisense), which effectively depletes RLIP, has tremendous potential to target vital nodes of importance in the survival, progression, and therapeutic resistance of melanomas.
References:
1 Spain L, Larkin J, Turajlic S. 2020. New survival standards for advanced melanoma. Br J Cancer 122(9):1275-1276. doi: 10.1038/s41416-020-0738-5. Epub 2020 Feb 17. PMID: 32063602; PMCID: PMC7188675.
2 Melanoma Research Alliance. Understanding Melanoma Staging. https://www.curemelanoma.org/about-melanoma/melanoma-staging/.
3 Singhal SS, Horne D, Singhal J, et al. 2021. Activating p53 function by targeting RLIP. Biochimica et Biophysica Acta (in-press). PMID: 33460725.
4 Singhal SS, Mohanty A, Kulkarni P, et al. 2021. RLIP depletion induces apoptosis associated with inhibition of JAK2/STAT3 signaling in melanoma cells. Carcinogenesis 42:742-752. PMID: 33623991.
Link:
Public and Technical Abstracts: Targeting the Oncoprotein RLIP as Novel Therapy for Melanoma
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
