Lupus
The LRP Presents the Fiscal Year 2021 Impact Award Recipients



Posted July 28, 2023
Anca Askanase, M.D., M.P.H., Columbia University Medical Center
Almost all patients with systemic lupus erythematosus (SLE) experience joint problems and arthritis, with nearly 12% suffering permanent joint damage as a result of the disease. However, the wide variety of SLE joint problems and the limitations of existing assessment tools make it difficult to identify joint issues in clinical care and SLE clinical trials. Clinicians need a robust, easy-to-use imaging technique to diagnose and monitor arthritis in SLE patients. With the support of a fiscal year 2021 (FY21) Impact Award, Dr. Anca Askanase is collaborating with Dr. Andreas Hielscher and his bioengineering team to further develop a prototype imaging system that uses near-infrared lasers and light detectors to evaluate finger joints in SLE patients. The system, which could take the form of an ergonomic glove, will allow the investigators to image the bones, joints, and tendons in the hands of study participants, both in the office and at home. The results will be compared against other arthritis assessment methods, thereby allowing the researchers to evaluate the tool's reliability for diagnosing and monitoring arthritis progress in SLE patients. Furthermore, the investigators will use these results to better characterize SLE-associated arthritis by examining the presence of proteins, called cytokines, that are associated with joint inflammation. The goal is to create a portable, wearable, imaging device that permits real-time monitoring of SLE arthritis to improve patient care and outcomes.
 (A) Dr. Anca Askanase's Columbia University Medical Center team
(B) The prototype imaging system for evaluating finger joints in SLE patients
(C) Dr. Andreas Hielscher's New York University's bioengineering team
(D) Dr. Anca Askanase evaluating a patient
(E) Dr. Andreas Hielscher.
(A) Dr. Anca Askanase's Columbia University Medical Center team
(B) The prototype imaging system for evaluating finger joints in SLE patients
(C) Dr. Andreas Hielscher's New York University's bioengineering team
(D) Dr. Anca Askanase evaluating a patient
(E) Dr. Andreas Hielscher.
Link:
Public and Technical Abstracts: Evaluation of Lupus Arthritis Using Novel Technologies
Karen H. Costenbader, M.D., M.P.H. and Deepak A. Rao, M.D., Ph.D., Brigham and Women's Hospital
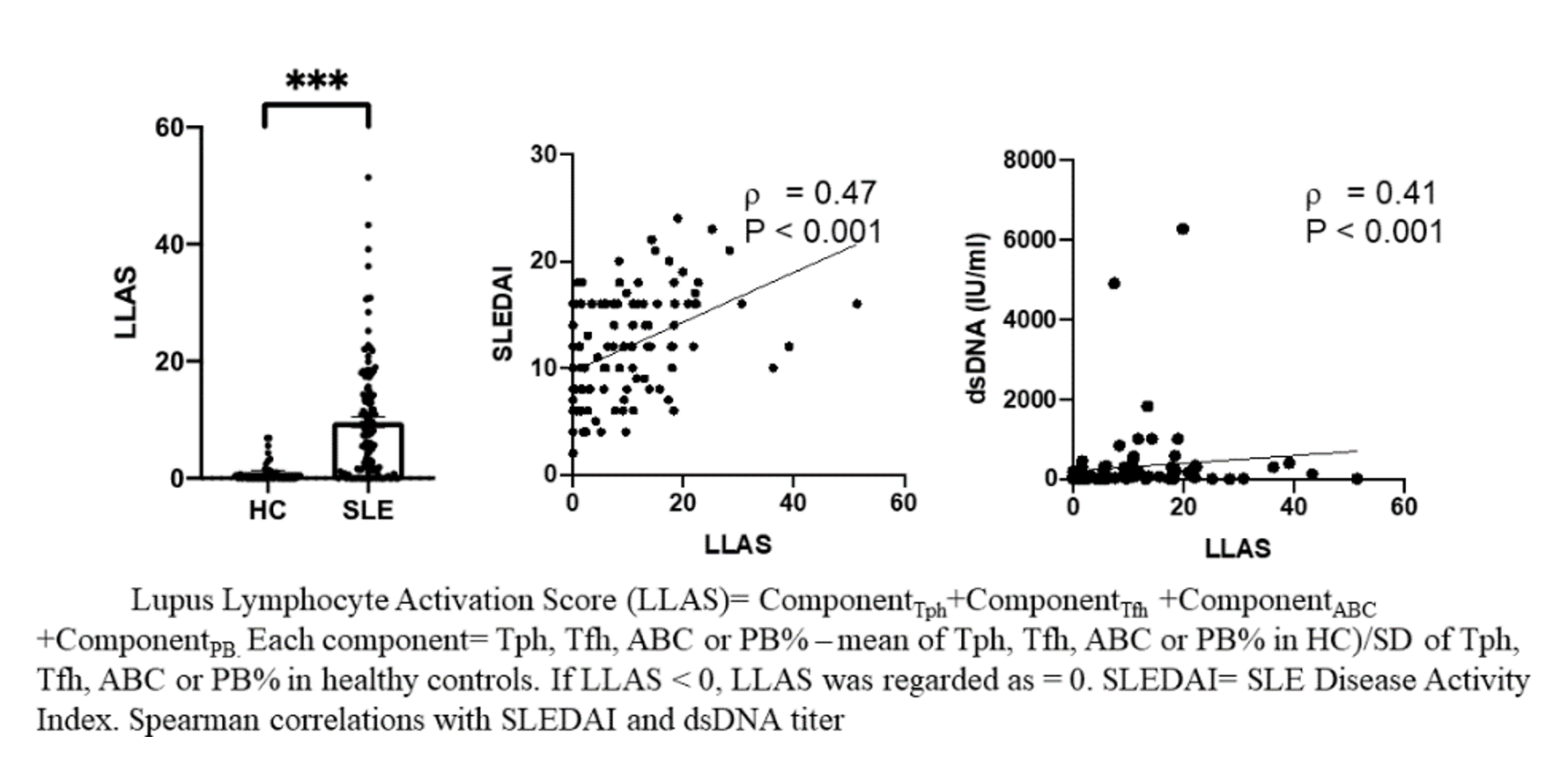
The symptoms of systemic lupus erythematosus (SLE) are difficult to identify in the early stages, often resulting in delayed diagnoses that lead to poorer outcomes for patients. The ability to identify and treat lupus earlier would represent a significant breakthrough in patient care. Seeking a way to do that, Drs. Costenbader's and Rao's team is studying four types of specialized white blood cells called lymphocytes - T peripheral helper cells, T follicular helper cells, age-associated B cells, and plasmablasts - in patients who may be developing SLE. With the support of an FY21 Impact Award, Drs. Costenbader and Rao will investigate whether higher-than-average percentages of these lymphocytes can be used to identify the presence of SLE in patients with early and non-specific symptoms. They will use cutting-edge immunophenotyping and machine learning to examine blood samples from 40 SLE patients to characterize cells and profiles associated with the various clinical symptoms of SLE, and also with the production of two proteins called autoantibodies and cytokines. The team expects the results of their 3-year study will not only help clinicians diagnose SLE earlier, but also reveal whether novel drug treatments should target a specific lymphocyte population or focus instead on reducing overall lymphocyte production in SLE.
 The Costenbader and Rao Lupus Research Team at the Brigham and Women's Hospital Division of Rheumatology, Inflammation and Immunity. From left to right: Takanori Sasaki, M.D., Ph.D. (post-doctoral fellow), Kathryne Marks, Ph.D. (post-doctoral fellow), Ifeoluwakiisi Adejoorin (research technician), Eilish Dillon (research assistant), Emily Oakes (research assistant), Karen Costenbader, M.D., MPH (PI), Alice Horisberger, M.D. (post-doctoral fellow), and Deepak Rao, M.D., Ph.D. (co-PI).
The Costenbader and Rao Lupus Research Team at the Brigham and Women's Hospital Division of Rheumatology, Inflammation and Immunity. From left to right: Takanori Sasaki, M.D., Ph.D. (post-doctoral fellow), Kathryne Marks, Ph.D. (post-doctoral fellow), Ifeoluwakiisi Adejoorin (research technician), Eilish Dillon (research assistant), Emily Oakes (research assistant), Karen Costenbader, M.D., MPH (PI), Alice Horisberger, M.D. (post-doctoral fellow), and Deepak Rao, M.D., Ph.D. (co-PI).
Link:
Public and Technical Abstracts: Dissecting Lymphocyte Dynamics to Identify Early Lupus
C. Garrison (Garry) Fathman, M.D., Stanford University Medical School
 Dr. Linda Yip and Dr. Garry Fathman
Dr. Linda Yip and Dr. Garry Fathman
Regulatory T cells (Tregs) are specialized white blood cells that prevent the body's immune system from attacking healthy tissues and organs. Researchers believe that defects in Tregs may contribute to the development of systemic lupus erythematosus (SLE), although the role that Tregs play in the pathology of SLE remains unclear. Dr. Fathman proposes to use his FY21 Impact Award to determine whether a common defect in the Tregs of SLE patients can be corrected using precise amounts of a tailor-made drug called a protein drug conjugate (PDC). Dr. Fathman hypothesizes that the Treg defect is due to a signaling malfunction in a protein – the interleukin-2 receptor (IL-2R) – found in Tregs from patients with autoimmune diseases.1 Delivering the PDC via the IL-2R may restore their function, thereby causing the Tregs to begin regulating the immune system correctly. Dr. Fathman's lab has previously shown that a mouse version of the PDC is therapeutically effective in various mouse models of autoimmunity. The 2-year research project will involve developing a human version of the PDC; developing a biomarker panel that can be used to identify the IL-2R defect in SLE patients; and testing whether the PDC can restore function to the IL-2R receptor signaling in Tregs from SLE patients and thereby enable the Tregs to function correctly. If successful, the research could lead to the development of PDCs that are suitable for the clinical treatment of SLE.
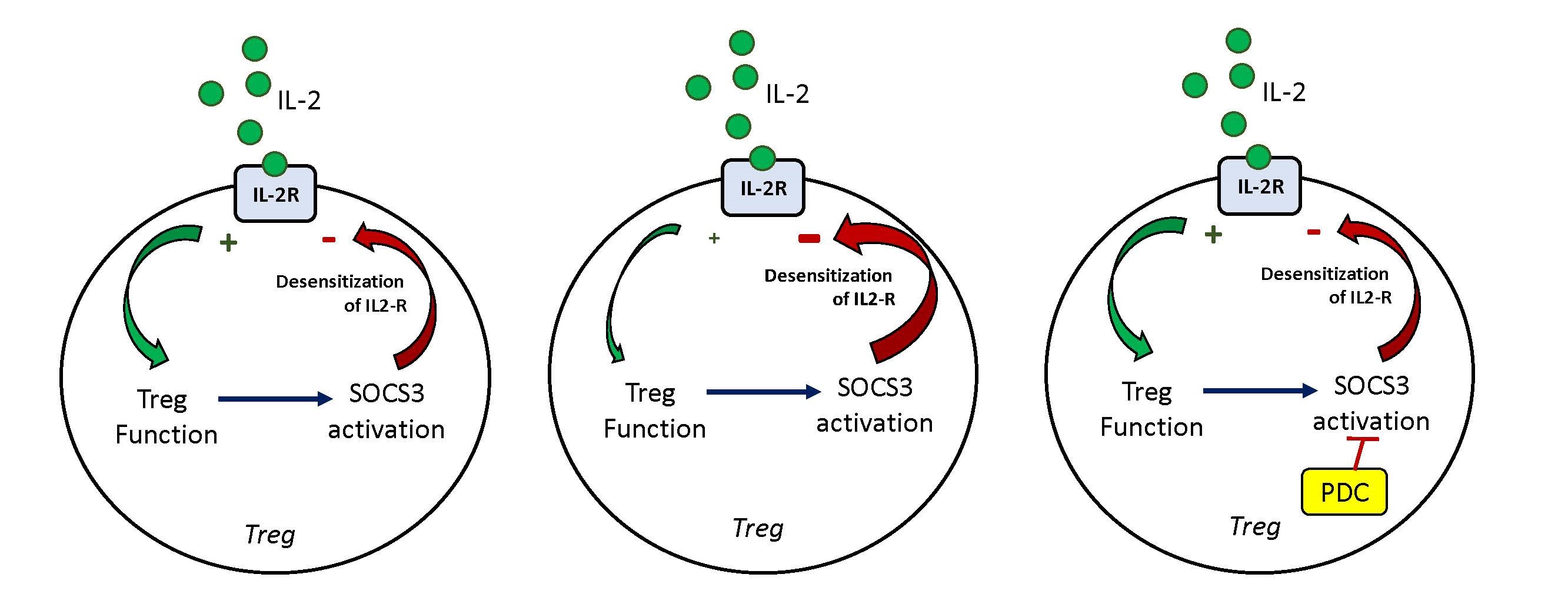 (Left) In a healthy individual, IL-2 binds to IL 2 receptors (IL-2R) to stimulate Treg function. SOCS3 is activated and results in the desensitization of the IL-2R.
(Left) In a healthy individual, IL-2 binds to IL 2 receptors (IL-2R) to stimulate Treg function. SOCS3 is activated and results in the desensitization of the IL-2R.
(Center) In patients with systemic lupus erythematosus (SLE) and autoimmune diseases, a defect in the Tregs of these patients results in the increased desensitization of the IL 2R and reduced Treg function.
(Right) In patients treated with the protein drug conjugate (PDC), the PDC targets the SOCS3 complex to reduce desensitization of the IL-2R. This allows for the restoration of Treg function in patients with SLE and autoimmune diseases.
Reference:
1Fathman, CG, Yip L, Gomez-Martin D, et al. 2022. How GRAIL controls Treg function to maintain self-tolerance. Frontiers in Immunology 13:1046631. https://doi.org/10.3389/fimmu.2022.1046631
Jason Knight, M.D., Ph.D., University of Michigan
 Dr. Jason Knight
Dr. Jason Knight
Approximately one-third of patients with systemic lupus erythematosus (SLE) have proteins in their blood called antiphospholipid antibodies, which increase their risk of experiencing blood clots and pregnancy loss due to a condition called antiphospholipid syndrome (APS). Furthermore, half of those patients are likely to acquire other harmful conditions in the brain, heart, and kidneys due to the failure of the blood vessels that supply those organs. Dr. Knight seeks to use his FY21 Impact Award to better understand what causes blood vessels in APS patients to fail over time. He and his colleagues will test the hypothesis that constant attack by antiphospholipid antibodies - which are among the most common antibodies in SLE patients - weakens the cells that line the inner walls of blood vessels, called endothelial cells. The team will use advanced molecular profiling to identify the genes and proteins that cause blood vessel failure in easily accessible skin tissue. The team hopes that by understanding why skin blood vessels fail, they will also learn something about why the same thing is happening to the blood vessels that supply internal organs. This 3-year study, the first of its kind to examine the actual blood vessel cells of APS patients, should yield novel biomarkers and new drug candidates for clinical trials.
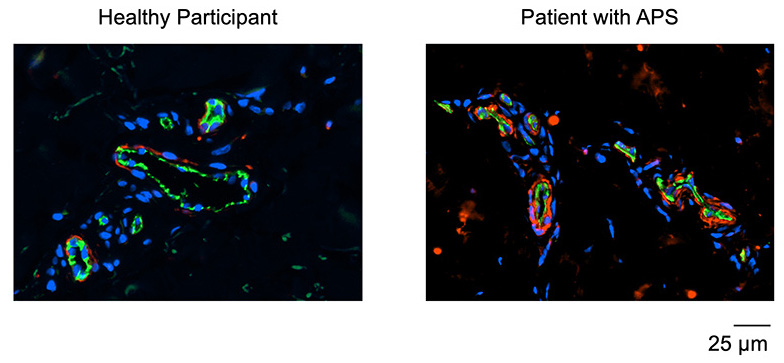 Immunofluorescence microscopy of skin biopsies. Cell nuclei are stained blue. The green staining highlights microscopic blood vessels caught in cross-section. The red staining is for an abnormal protein that Dr. Knight's team has found to be increased in patients with antiphospholipid syndrome (APS). Research is underway to further define the properties of that protein and the extent to which it may be a treatment target.
Immunofluorescence microscopy of skin biopsies. Cell nuclei are stained blue. The green staining highlights microscopic blood vessels caught in cross-section. The red staining is for an abnormal protein that Dr. Knight's team has found to be increased in patients with antiphospholipid syndrome (APS). Research is underway to further define the properties of that protein and the extent to which it may be a treatment target.
Link:
Public and Technical Abstracts: Endothelial Cells and the Origins of Antiphospholipid Vasculopathy
Tamara Nowling, Ph.D., Medical University of South Carolina
 Dr. Tamara Nowling's Lupus Research Group (front row, left to right: Sandy Mungaray and Aaron Angerstein; back row, left to right: Beth Wolf, Ph.D., Tammy Nowling, Ph.D., Rick Drake, Ph.D., Danielle Beaufort, M.S., and Jim Oates, M.D.)
Dr. Tamara Nowling's Lupus Research Group (front row, left to right: Sandy Mungaray and Aaron Angerstein; back row, left to right: Beth Wolf, Ph.D., Tammy Nowling, Ph.D., Rick Drake, Ph.D., Danielle Beaufort, M.S., and Jim Oates, M.D.)
Kidney disease, also called nephritis, is the most severe complication associated with systemic lupus erythematosus (SLE), affecting two-thirds of SLE patients. One of the characteristics of SLE-related nephritis is elevated levels of two lipids, lactosylceramide (LacCer) and hexosylceramide (HexCer), in the urine of SLE patients with nephritis. In previous research, Dr. Nowling determined that pre-treatment levels of LacCer and HexCer in patients who ultimately did not respond to standard nephritis treatment were much higher than in those patients who did respond to treatment. She more recently demonstrated that the pre-treatment N-glycome (pattern of protein N-linked glycosylation or N-glycans), was significantly altered in the urine of patients who did not respond to standard nephritis treatment. Now, with the help of her FY21 Impact Award, Dr. Nowling will build on these findings by conducting a study of LacCer and HexCer levels and the N-glycome in urine samples collected from SLE patients with and without nephritis, as well as from healthy subjects with the goal of developing a biomarker panel to better predict which patients will respond to standard nephritis treatments before beginning treatment. This knowledge will help clinicians identify patients who require more aggressive therapies and closer monitoring, leading to more informed treatment decisions and improved patient outcomes. Dr. Nowling's group is also investigating how altering LacCer and HexCer levels impacts the function of kidney cells and contributes to disease progression.
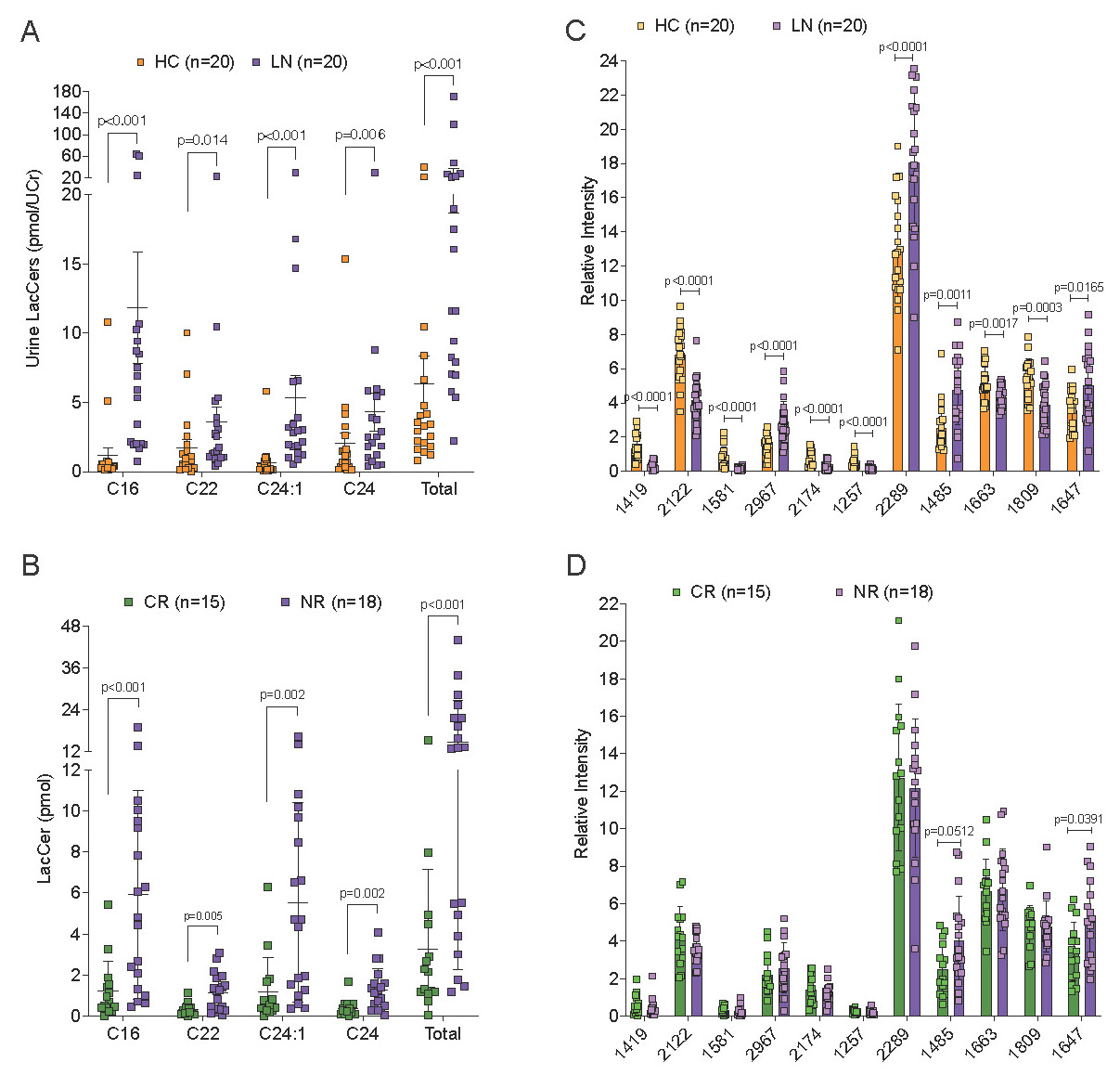 Lactosylceramide (LacCer) levels are significantly elevated (A) and the N-glycome is significantly altered (C) in the urine of lupus nephritis (LN) patients (purple) compared to healthy control (HC) subjects (orange). Graph in (C) shows 11 of 72 N-glycans that are significantly different between LN and HC. (B) Urine LacCer levels prior to treatment are highest in LN patients that did not respond (NR, purple) compared to those that completely responded (CR, green) to standard therapy. (D) Two N-glycans in urine are significantly higher prior to treatment in NR LN patients (purple) compared to CR LN patients (green). A and C, urine samples were collected during active disease from LN patients. B and D, therapeutic response was determined at 12 months post-treatment.
Lactosylceramide (LacCer) levels are significantly elevated (A) and the N-glycome is significantly altered (C) in the urine of lupus nephritis (LN) patients (purple) compared to healthy control (HC) subjects (orange). Graph in (C) shows 11 of 72 N-glycans that are significantly different between LN and HC. (B) Urine LacCer levels prior to treatment are highest in LN patients that did not respond (NR, purple) compared to those that completely responded (CR, green) to standard therapy. (D) Two N-glycans in urine are significantly higher prior to treatment in NR LN patients (purple) compared to CR LN patients (green). A and C, urine samples were collected during active disease from LN patients. B and D, therapeutic response was determined at 12 months post-treatment.
Joshua Ooi, Ph.D., Monash University
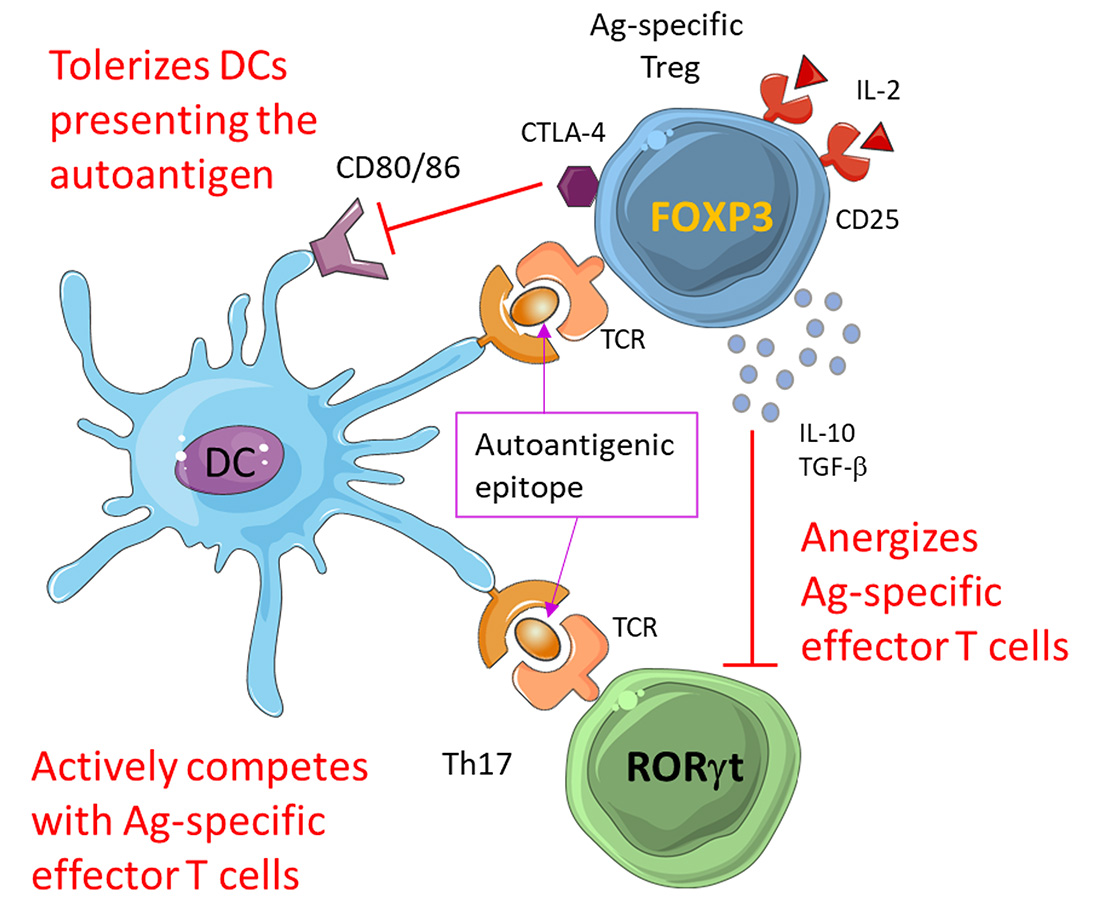 Antigen-specific Tregs are potent suppressors of autoreactivity because they induce immunological tolerance in dendritic cells (DCs) presenting the autoantigen, cause pro-inflammatory T cells to respond to the Smith antigen, and outcompete antigen-specific effector T cells for binding of the autoantigen.
Antigen-specific Tregs are potent suppressors of autoreactivity because they induce immunological tolerance in dendritic cells (DCs) presenting the autoantigen, cause pro-inflammatory T cells to respond to the Smith antigen, and outcompete antigen-specific effector T cells for binding of the autoantigen.
For people experiencing the more severe manifestations of systemic lupus erythematosus (SLE) such as kidney disease (nephritis), one of the symptoms most frequently associated with their illness is an immune response against a type of protein called the Smith (Sm) antigen. Dr. Ooi and his team have developed and patented
 Dr. Josua Ooi and his research team
Dr. Josua Ooi and his research team
Link:
Public and Technical Abstracts: Targeted Regulatory T Cells to Treat SLE
Eric Sebzda, Ph.D., Wayne State University
While researchers know that a subset of white blood cells, called B and T cells, interact with environmental factors to cause systemic lupus erythematosus (SLE), the process by which these three components come together to initiate disease is not understood. Dr. Sebzda's team found that the removal of a protein involved in the control of molecules located on the membranes of B cells called homing receptors was sufficient to promote SLE in genetically modified mouse models. Preliminary data suggests that B cells that are typically confined to body cavities help reduce the immunological tolerance of T cells to prevent immunity directed against harmless environmental molecules. By altering the location of these B cells so that they can no longer interact with environmental factors found in the lungs and guts, an immune-suppressive function is eliminated, leading to SLE. Dr. Sebzda will use his FY21 Impact Award to test if this model explains how B cells, T cells, and environmental factors converge to cause SLE. Results from these studies may identify a new mechanism of immune tolerance. In turn, this work will enable the development of therapeutic interventions that prevent a breakdown in interactions between T cells and B cells, leading to SLE. This includes a population of ethnic Chinese patients that appear to be particularly sensitive to this form of SLE.
Link:
Public and Technical Abstracts: Identifying a KLF2-Dependent B-Cell Mechanism Responsible for Lupus
Last updated Friday, December 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
