Lupus
The LRP Presents the Fiscal Year 2021 Idea Award Recipients



Posted June 15, 2023
Benjamin Chong, M.D., University of Texas, Southwestern Medical Center at Dallas
For patients with cutaneous lupus erythematosus (CLE), determining how they respond to treatments relies on serial skin examination assessments. Advances in provider assessment tools have helped providers measure skin disease severity in CLE; however, there is a gap in tools that help represent how CLE patients feel about their skin condition. The need for representation of the patient perspective is significant in addressing treatment response, as provider perceptions do not always corroborate patient experience. To address this challenge, Dr. Chong and his team developed the Cutaneous Lupus Erythematosus Quality of Life (CLEQoL) assessment tool. The questionnaire is comprised of five topic sections focusing on emotions, symptoms, functioning, photosensitive, and body image/cosmetic effects. Feedback from patients provides a comprehensive understanding of the impact of CLE on patient needs. Through a fiscal year 2021 (FY21) Idea Award, Dr. Chong will evaluate the reliability and validity of the CLEQoL in a larger multi-center group of CLE patients. He will continue to evaluate how CLEQoL scores will adjust when variance in disease severity is acknowledged. The study will compare CLEQoL patient reported scores with physician assessment scores to determine correlation. If successful, the addition of the CLEQoL matrix will help better treat skin disease in CLE patients and ultimately improve quality of life for individuals with lupus. It can serve as a useful tool to help represent patient perspectives on CLE, which can ultimately be used to help identify effective therapies. Upon conclusion of this award, Dr. Chong expects to use the gained information toward obtaining U.S. Food and Drug Administration approval for the patient-oriented outcome measure.
 Dr. Benjamin Chong (sixth from left)
Dr. Benjamin Chong (sixth from left)
Michael Crackower, Ph.D., Ventus Therapeutics U.S., Inc.
 Dr. Michael Crackower
Dr. Michael Crackower(Photo Provided)
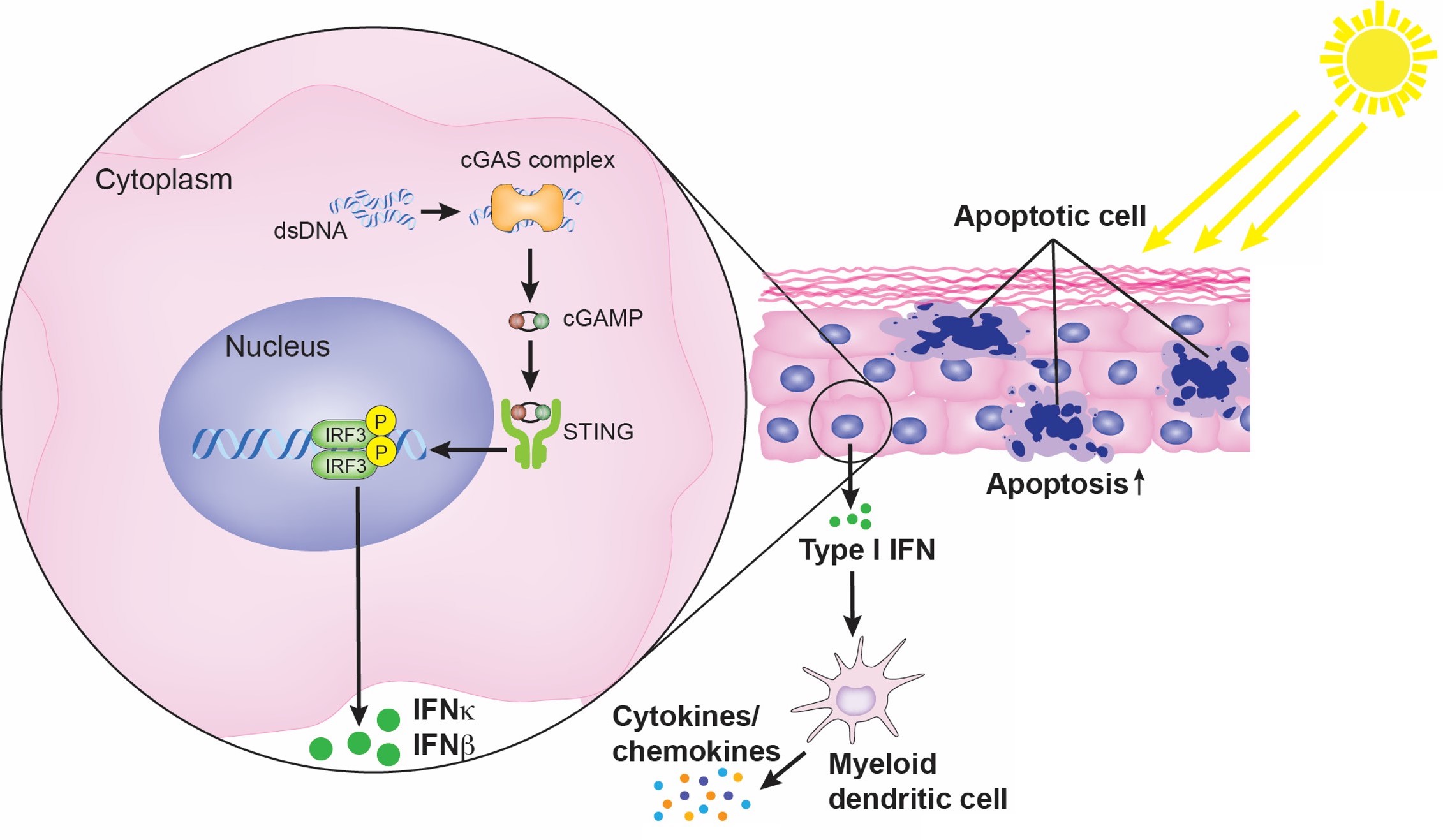 Figure 1: UVB exposure exacerbates skin inflammation in SLE by triggering activation of the cGAS-STING pathway and the release of pro-inflammatory cytokines. (Figure Provided)
Figure 1: UVB exposure exacerbates skin inflammation in SLE by triggering activation of the cGAS-STING pathway and the release of pro-inflammatory cytokines. (Figure Provided)
Approximately 70% of patients with systemic lupus erythematosus (SLE) develop hypersensitivity to ultraviolet B (UVB) wavelengths produced by the sun. Exposure can produce rashes or lesions and induce progression of SLE in regions not exposed to UV, causing systemic manifestations. Post-UV damage is correlated to the activation of the enzyme cGAS and interferons responsible for inflammation in lupus patients. With an FY21 Idea Award, Dr. Crackower seeks to better understand how cGAS is involved in UV-induced inflammation. He and his team will assess the effects of inhibiting cGAS in lupus models and explore the protein in skin cells after UV exposure. The proposed research will help generate important data for future development of therapeutics treating inflammation in lupus patients. For example, the results of this study may lead to the development of a topical solution, such as a sunscreen, that lupus patients could use prior to UV exposure. If successful, Dr. Crackower and his team plan to continue their research into UV-induced inflammation, focusing on U.S. Food and Drug Administration Investigative New Drug-enabling studies leading up to a phase 1 first-in-human clinical trial.
Link:
Public and Technical Abstracts: Understanding the Role of cGAS in SLE and UVB-Induced Inflammation
Linda Hiraki, M.D., Sc.D., The Hospital for Sick Children
 Dr. Michael Wilson
Dr. Michael Wilson
(Photo Provided)
 Dr. Eleanor Pullenayegum
Dr. Eleanor Pullenayegum
(Photo Provided)
 Dr. Cynthia Guidos
Dr. Cynthia Guidos
(Photo Provided)
 Dr. Linda Hiraki
Dr. Linda Hiraki(Photo Provided)
Lupus is an autoimmune disease resulting from inherited genetic factors as well as environmental variables. Approximately 20% of people with lupus are diagnosed in childhood or adolescence. Lupus symptoms often vary, resulting in a wide range of disease courses and therapeutic responses. Few lupus studies have analyzed gene expression before and after treatment initiation, along with clinical changes over time. To address the gap in knowledge, Dr. Linda Hiraki is working toward identifying how gene expression and an abundance of immune cells correlates with disease manifestations and changes in lupus activity and progression. With an FY21 Lupus Research Program Idea Award, the team, which includes experts in immunology (Dr. Cynthia Guidos' lab), biostatistics (Dr. Eleanor Pullenayegum's lab) and genomics (Dr. Michael Wilson's lab), will explore the hypothesis that specific immune cells and treatment-related gene expression profiles are associated with clinical disease characteristics and disease activity. They will further examine how gene expression relates to the differences in therapy response. The research team's work will provide valuable insights into the complex biology of lupus, and the findings may inform future therapeutic development. Collectively, these discoveries will help launch new approaches for lupus treatments, helping improve care and the quality of life for patients with lupus.
Theresa Lu, M.D., Ph.D., Hospital for Special Surgery
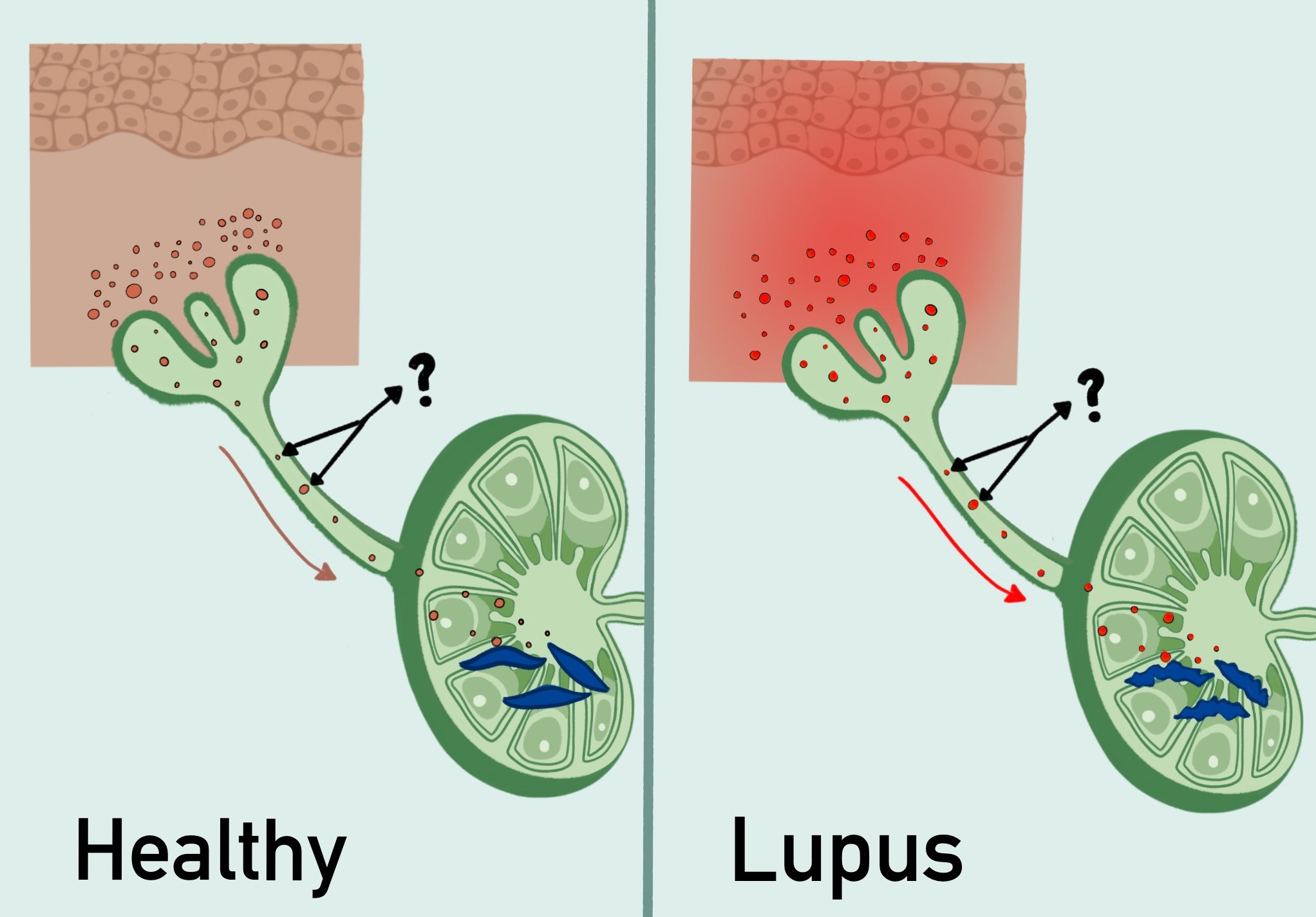 Figure 2: Schematic of the skin-lymph node axis in health and lupus. In health, molecules (dull red) travel from skin to lymph nodes via lymphatic vessels that then act on fibroblasts (blue) to control immunity. In lupus, the skin is inflamed, and the molecules from skin now cause lymph node fibroblasts to behave differently, leading to autoimmunity. Dr. Lu and team will be examining the differences in the nature of the molecules between health and disease.
Figure 2: Schematic of the skin-lymph node axis in health and lupus. In health, molecules (dull red) travel from skin to lymph nodes via lymphatic vessels that then act on fibroblasts (blue) to control immunity. In lupus, the skin is inflamed, and the molecules from skin now cause lymph node fibroblasts to behave differently, leading to autoimmunity. Dr. Lu and team will be examining the differences in the nature of the molecules between health and disease.
Patients with systemic lupus erythematosus (SLE) develop inflammatory skin lesions as a direct result of sunlight exposure, due to an increased sensitivity to ultraviolet radiation (UVR). Photosensitive skin responses can cause systemic disease flare ups and increased circulating autoantibodies, which deposit in organs, furthering injury. Scientific advances have provided insight on the cause of photosensitivity; however, response mechanisms are still not well understood. With an FY21 Idea Award, Dr. Lu will help address this need by further exploring the biological mechanisms of lupus disease. She and her colleagues will test the hypothesis that photosensitivity is linked to systemic flares because excess interferons and other signals from SLE activate inflammation upon UVR exposure, limiting fibroblast cell function and reducing autoimmune responses. Through their research strategy, Dr. Lu and her team will identify the nature of signals in lupus that are transmitted from healthy skin to lymph nodes and further examine how fibroblasts' immune-regulatory functions are altered by lupus skin-derived signals. This study will provide insight on the biological mechanisms involved and address the connection between photosensitivity and systemic flares. The data Dr. Lu will collect from this study will be used to expand understanding, gain a better understanding of preventative measures, and help develop advanced therapies for UVR lesions in SLE patients for improvement in quality of life.
 Dr. Theresa Lu's research team
Dr. Theresa Lu's research team
(Photo Provided)
Link:
Public and Technical Abstracts: Photosensitivity and Lymph Node Immune Responses in Lupus
Keisa Mathis, Ph.D., University of Texas, Health Science Center, Fort Worth
 Dr. Keisa Mathis
Dr. Keisa Mathis(Photo Provided)
Chronic inflammation is a life-threatening symptom of systemic lupus erythematosus (SLE), which increases the potential for long-term inflammation in organs like the kidney and the skin, contributing to chronic pain and fatigue. While current therapies for SLE help suppress symptoms, they often result in adverse side effects and impact the quality of life for SLE patients. Novel and safe therapeutics options are desperately needed to help combat uncontrollable inflammation to provide relief and deliver better outcomes. With an FY21 Idea Award, Dr. Mathis seeks to better understand the biological impact of SLE while further investigating the pathobiology of end organ injury. She and her colleagues will further explore the hypothesis that splenic nerve stimulation will reduce the production and release of pro-inflammatory cytokines from splenic immune cells, which would subside renal inflammation, reducing stress and preventing the development of renal injury in SLE patients. The team will induce splenic nerve activity with the use of ultrasound technology in an SLE animal model to test injury-prevention capabilities and reduce chronic inflammation. Increased nerve stimulation is expected to reduce inflammation throughout the entire body, suppressing pain receptors and minimizing long-term impact. Understanding how splenic nerve activity can impact inflammation response can lead to better therapeutic outcomes.
Link:
Public and Technical Abstracts: Preventing Renal Injury in Lupus Nephritis via Ultrasound Therapy
Last updated Friday, December 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
