Lupus



Posted March 1, 2022
Richard Looney, M.D., University of Rochester Medical Center
 Dr. Richard Looney
Dr. Richard Looney
About 50% of adults and 80% of children with lupus will develop lupus nephritis, and between 10% to 30% of those will go on to acquire end-stage renal disease. The current lupus treatment paradigm of high-dose steroids and immunosuppressive medications has been standard for the last four decades; however, these medications often turn the acute inflammation and rapid progression of renal disease into chronic inflammation and slow, but do not stop, the still inevitable progression of end-stage renal disease. Drugs that target senescent cells (senolytic drugs) are used to rejuvenate tissues, and have the potential to improve kidney function in patients with renal disease. Senescent cells are abnormal cells that can interfere with the normal functions of surrounding cells. Accumulation of these cells is associated with poor function in multiple organs, including the heart, blood vessels, kidneys, muscles, bones and brain. Senescent cells normally accumulate as people age; however, they can also be induced by various types of stress.
Many senolytic drugs are already approved for human use and many cells expressing senescence have been identified in other types of chronic kidney disease, however these studies did not include patients with lupus. Dr. Richard Looney and his research team have previously identified bone marrow mesenchymal stem cells (BM-MSC) from patients with systemic lupus erythmatosis (SLE) that exhibit signs of senescence. With a Fiscal Year 2017 Lupus Research Program Concept Award, Dr. Looney aimed to evaluate the accumulation of senescent cells in lupus target tissue and to see if the senescent cells can be targeted by existing senolytic drugs.
Dr. Looney proposed that senescent tissues are increased in SLE target tissues and contribute to organ dysfunction, decreased activity and poor long term outcomes. He also hypothesized that senolytic drugs can selectively eliminate senescent cells from patients with SLE and rejuvenate these tissues, improve organ function, and promote disease dormancy. To assess these hypotheses, a variety of senolytic drugs were tested in vitro (in a test tube) on BM-MSC from SLE patients and from lupus model (NZB/W) mice. The kidney tissue of SLE patients and NZB/W mice was also examined for markers of senescence.
To assess the effect of senolytic drugs on human BM-MSC, several senolytic drugs were screened. The team found that the drug FOXO4-DRI had induced the most significant cell death (apoptosis) and had the least effect on the survival of healthy cells. This drug was further tested and applied to SLE BM-MSCs. FOXO4-DRI inhibited SLE BM-MSC survival and induced apoptosis (normal cell death) more than 2 fold, suggesting that FOXO4-DRI can induce apoptosis of senescent SLE BM-MSCs. FOXO4-DRI was also tested in NZB/W mice BM-MSCs with similar apoptosis-inducing results.
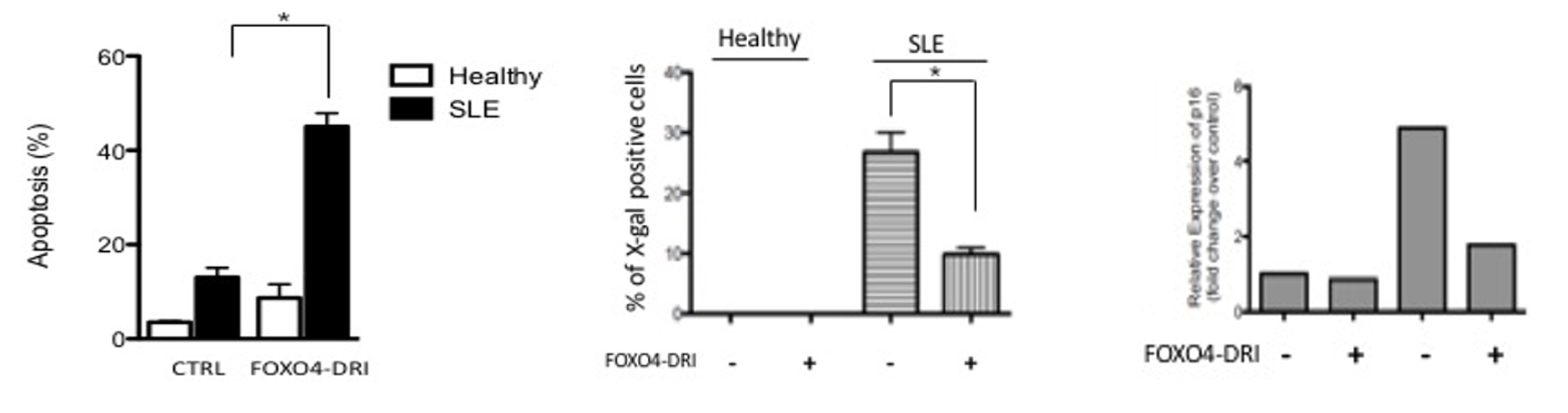 FOXO4-DRI is able to eliminate senescent cells in tissue culture. We have previously shown that bone marrow mesenchymal stromal cells for SLE patients have a senescent phenotype. Treatment with a new senolytic drug (FOXO4-DRI) induces apoptosis (left figure) and eliminates SLE mesenchymal stromal cells expressing markers of senescence (middle figure senescence associated beta galactosidase and right figure p16).
FOXO4-DRI is able to eliminate senescent cells in tissue culture. We have previously shown that bone marrow mesenchymal stromal cells for SLE patients have a senescent phenotype. Treatment with a new senolytic drug (FOXO4-DRI) induces apoptosis (left figure) and eliminates SLE mesenchymal stromal cells expressing markers of senescence (middle figure senescence associated beta galactosidase and right figure p16).
Moving on to assessing SLE kidney tissue, the team chose to use FOXO4 as an additional senescence marker because their chosen drug, FOXO4-DRI, modulates its expression. Kidneys from NZB/W and age-matched control mice were harvested and analyzed for the presence of the senescence markers. The results suggest that NZB/W mice have increased expression of senescence markers such as FOXO4, rH2AX, and p16, and that senescent cells can be targeted in the kidney of patients with lupus.
Dr. Looney and his team have documented that BM-MSC cells from patients with SLE or lupus model mice are a mix of senescent and healthy cells, and that treatment with FOXO4-DRI can selectively induce apoptosis in senescent cells, allowing the healthy cells to continue to grow and multiply. The team has also documented markers of senescence in kidneys of lupus prone mice. These findings suggest that use of senolytic drugs in lupus may selectively eliminate senescent cells in kidneys and other tissues. The research team hopes that senolytic drugs can be used to change this treatment paradigm and improve the quality of life for individuals with SLE.
Link:
Public and Technical Abstracts: Therapeutic Targeting of Senescent Cells in Lupus
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
