Lung Cancer



Posted July 23, 2021
Fumito Ito, M.D., Ph.D., Roswell Park Comprehensive Cancer Center

Dr. Fumito Ito
Immune checkpoint inhibition (CPI) has dramatically improved the therapeutic regimen available to patients with advanced non-small cell lung cancer (NSCLC), yet not all patients experience long-lasting clinical benefits. Compounding this division in therapeutic benefit are factors such as possible severe immune-related adverse effects brought on by CPI, as well as the high costs associated with therapy. Ultimately, to maximize CPI’s utility, while mitigating risk and cost to patients for whom CPI is unlikely to be helpful, the development of a biomarker to predict clinical benefit in NSCLC patients is required. In the lung, monitoring therapeutic benefit by image-based metrics has thus far proven only modestly effective, and serial biopsy presents a highly invasive burden on the patient. Optimally, predictive biomarkers of CPI benefit would incorporate elements of high specificity and minimal invasiveness, such as could be achieved through a routine blood draw. This is precisely the effort being undertaken by Dr. Fumito Ito, with the assistance of a fiscal year 2018 Lung Cancer Research Program, Investigator-Initiated Translational Research Award.
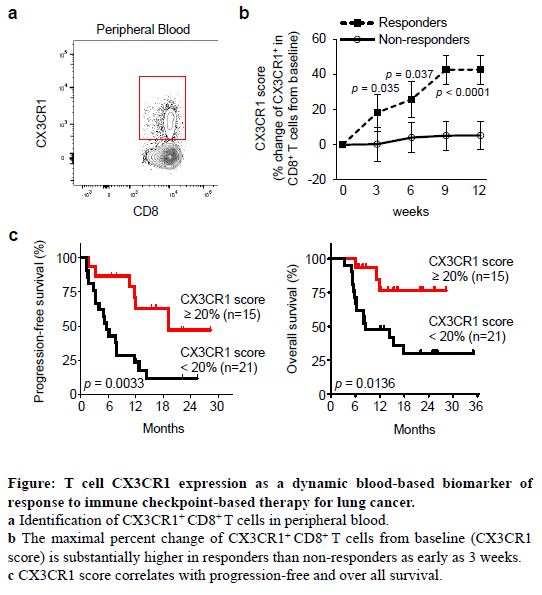
Dr. Ito’s work builds on the observation that CPI therapy increases the frequency of effector CD8+ T-cells in the blood stream that express the chemokine receptor CX3CR1. Importantly, the presence of CX3CR1+ CD8+ T-cells in the blood (peripheral T-cells) has been linked to clonal (highly similar), tumor-reactive T-cells within tumors in animal models, indicating CX3CR1+ CD8+ peripheral T-cells may serve as a surrogate for an active immune response to NSCLC. Armed with this information, Dr. Ito’s group is pursuing the usefulness of CX3CR1 as a peripheral blood-based biomarker to predict patient response and toxicities associated with CPI, and to distinguish responders from non-responders early in treatment.
The team is recruiting advanced NSCLC patients receiving the anti-PD-1 antibody, a common CPI therapy, from whom blood is drawn over the course of treatment. Measures of how these patients’ tumors respond to therapy are collected, and information on any toxic side effects are also monitored. T-cells are then isolated from the patient blood draws and analyzed for the abundance of CD8+ CX3CR1+ T-cells and their clonal similarity, as well as having a detailed evaluation of the genetic and immune profile performed.

Dr. Grace Dy
Early interim analyses of 36 patients (of a planned 108) from the study were recently included in work published in Nature Communications. Dr. Ito’s group evaluated each sample, comparing changes in the CX3CR1+ subset of peripheral T-cells at each post-treatment collection, to the baseline level for each patient prior to treatment in collaboration with the Roswell Park Thoracic Oncology team, led by Dr. Grace Dy. The resulting percentage change over time is called the “CX3CR1 score,” where a 15% change from baseline is a score of 15, and so on. In these early results, the team determined that a CX3CR1 score of at least 20 was associated with a high accuracy for patient response to therapy at 3 (77.8%), 6 (77.8%), 9 (86.1%), and 12 (88.9%) weeks of therapy. Additionally, a CX3CR1 score greater than 20 at 12 weeks on treatment was associated with greater progression-free survival and, notably, overall survival. Importantly, the predictive value of Dr. Ito’s CX3CR1 score outperforms the typical measurement of treatment effectiveness, the tumor PD-L1 proportion score (TPS).
These early study results are likely to be further refined as Dr. Ito’s group digs deeper into the molecular profiles of these peripheral CX3CR1+ CD8+ T-cells, strengthening the predictive value of the emerging CX3CR1 score as a biomarker. Additional work to establish the CX3CR1 score as a predictor of a CPI-induced, immune-related serious adverse event will enhance the utility of this biomarker in the clinic. These developments are critically important for patients with advanced NSCLC, as they will enable a less invasive means by which to identify and monitor patients who will benefit from CPI and spare those who will not.
Publication:
Yamauchi T, Hoki T, Oba T, et al. 2021. T-cell CX3CR1 expression as a dynamic blood-based biomarker of response to immune checkpoint inhibitors. Nat Commun 12:1402.https://doi.org/10.1038/s41467-021-21619-0.
Links:
Roswell Park Researchers Identify New Biomarker of Response to Checkpoint Inhibitors
Last updated Thursday, November 13, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
