Lung Cancer



Posted November 3, 2021
Lynn Heasley, Ph.D., and Raphael Nemenoff, Ph.D., University of Colorado at Denver

Dr. Lynn Heasley

Dr. Raphael Nemenoff
Therapies that target the mutant EGFR gene and other oncogenic drivers of lung cancer can shrink tumors, delay disease progression, and improve patient overall survival. Despite these initial positive outcomes, most therapies fail to eradicate tumors entirely, resulting in residual disease, which spawns therapy-resistant recurrences. Furthermore, the initial effectiveness of these therapies can vary among distinct groups of patients, suggesting that specific biological modulators of the therapy may affect how profoundly a patient responds. With assistance from a Fiscal Year 2018 Lung Cancer Research Program Translational Research Partnership Award, Dr. Lynn Heasley and Dr. Raphael Nemenoff have been investigating very early molecular and cellular responses to targeted tyrosine kinase inhibitor (TKI) therapy to identify factors associated with deeper therapeutic response. Early results from the work provide evidence that suggests an immediate and robust inflammatory response to TKI therapy, measurable in clinical samples, is associated with better patient outcomes and may be exploited in order to design more durable co-treatment strategies.
While the study is still underway, the research team recently published a report highlighting the early effects of EGFR-targeting TKIs in NPJ Precision Oncology. Using a panel of EGFR-mutant lung cancer cells, the team investigated changes in active gene expression following a short time course (14-day) treatment with TKI therapy. All cells exhibited a very rapid enrichment of sets of genes associated with inflammation, notably those in the interferon alpha (IFNα) and interferon gamma (IFNγ) response pathways, though the intensity and duration of the enrichment varied. Follow-up evaluation of tumors established by these cells in mice confirmed that short-term TKI therapy activated both IFNα and IFNγ response pathways.
Common genes within the 297 associated with the IFNα and IFNγ response pathways include cytokines and chemokines that play important roles in communicating with immune cells in the local tumor microenvironment. Genes such as CXCL10 may facilitate an anti-tumor immune response, while genes like IL-6 may help establish a tumor-protective environment. Consistent with a differential response to TKI therapy as seen in the clinic, the lung cancer cells analyzed demonstrated variations in kinetics and intensity of chemokine and cytokine expression following treatment by TKI.
To begin translating these observations into the clinic, the team evaluated a small number of matched patient tumor biopsies corresponding to pre-treatment and approximately 2 weeks on treatment with an EGFR-targeted TKI. Detailed gene expression analysis revealed changes in IFNα and IFNγ response pathways in the patient samples on TKI therapy that mirror those seen in the cell lines, suggesting that TKI therapy induces an inflammatory response within patient tumors. Further evaluation showed that induction of the IFNγ response pathway significantly correlated with the patient’s time to progression (TTP), where a more robust induction of the IFNγ response predicted a longer TTP. Additionally, patients with a greater TTP (>12 months) had tumor biopsies with a higher number of T cells resident in the tumor microenvironment than did patients with a shorter TTP (0-8 months), suggesting a more anti-tumor microenvironment.
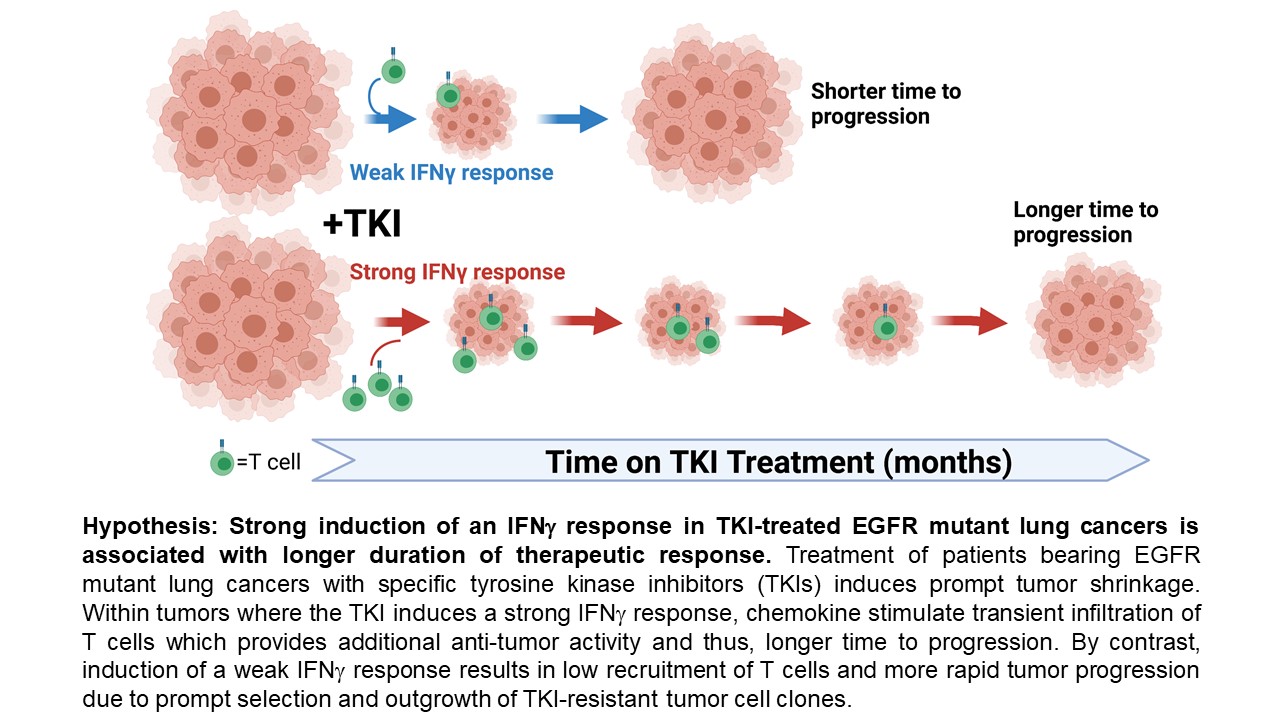
The research team’s efforts so far have indicated that TKI-induced inflammatory signaling causes a variable short-term depth of tumor response that may predict longer-term patient outcomes. Identifying the specific signaling molecules that mediate the depth of response through immune cell recruitment will likely open new pathways for the development of novel therapies in lung cancer. When used in combination, these new therapies would generate a more predictable, robust, and durable initial response to TKIs that could dramatically improve on important patient outcomes like time to progression.
 Heasley and Nemenoff lab groups (from left to right): Natalia Gurule, Dan Sisler, Lynn Heasley, Raphael Nemenoff, Andre Navarro, Emily Kleczko, and Teresa Nguyen.
Heasley and Nemenoff lab groups (from left to right): Natalia Gurule, Dan Sisler, Lynn Heasley, Raphael Nemenoff, Andre Navarro, Emily Kleczko, and Teresa Nguyen.
Publications:
Gurule NJ, McCoach CE, Hinz TK, et al. 2021. A tyrosine kinase inhibitor-induced interferon response positively associates with clinical response in EGFR-mutant lung cancer. NPJ Precision Oncology 5:41.https://doi.org/10.1038/s41698-021-00181-4.
Links:
Abstracts for Dr. Heasley
Abstracts for Dr. Nemenoff
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
