Lung Cancer



Posted November 20, 2020
Harold Varmus, M.D., Joan and Sanford I Weill Medical College of Cornell University
Small-cell lung cancer (SCLC) is an aggressive lung cancer that quickly develops resistance to treatment, leading to poor prognosis. It is known that SCLC is driven by genetic mutations of tumor suppressors retinoblastoma (RB) and tumor protein p53 (TP53); however, the origin and disease progression remains poorly understood. One potential method to better understand disease progression is to establish a stepwise model of the development of SCLC’s unique genotype as it transforms from its presumed parent cells. Small cells that compose SCLC are similar to naturally occurring but rare lung cells called pulmonary neuroendocrine cells (PNECs). In fact, PNECs have been speculated to be parent cells of SCLC cells. With support from a Fiscal Year 2016 (FY16) Lung Cancer Research Program Idea Development Award – Established Investigator (IDA-EI), Dr. Varmus and his team were able to demonstrate such a model by manipulating human embryonic stem cells (hESCs) into generating SCLC phenotypes.
First, the team induced hESCs to form PNEC-like cells by inhibiting NOTCH signaling, an important pathway regulating cell-cell interactions. They confirmed via immunohistochemistry and cell sorting assays that the PNEC-like cells differentiated from manipulated hESCs resulted from disrupting the NOTCH signaling pathway. However, single-cell RNA sequencing of these PNEC-like cells showed only ~8% of cells expressed characteristic PNEC markers. The team then inactivated the RB gene in NOTCH signaling-inhibited hESC cultures, which increased the percentage of cells expressing PNEC markers to ~38%. Dr. Varmus’ team then compared RNA profiles and found that these PNEC-like cells expressed several SCLC markers that more closely resembled those of human SCLC samples than non-PNEC-like cells derived from the same experimental pools. They also observed that the PNEC-like cells’ transcriptomes where both RB and TP53 expression levels were reduced had enhanced expression of genes associated with cell proliferation and apoptosis inhibition, which are hallmarks of tumor growth and progression.

Dr. Harold Varmus (2nd from right) and research team
To test whether PNEC-like cells could differentiate into SCLC-like tumors, the team subcutaneously injected these cells into immunodeficient mice and observed tumor growth on week seven. Mice injected with hESCs that were only NOTCH signaling-inhibited did not grow tumors larger than 250 mm3. Mice injected with hESCs where RB and TP53 were inactivated following inhibition of NOTCH signaling, on the other hand, grew visible tumors (~1cm in diameter) at seven weeks; immunohistological staining also showed these tumors to express several SCLC markers. The group reported that these SCLC-like tumors were slow in growth and potency, which could support studies on tumor development and progression. Furthermore, cells could be examined at different points of tumor progression when studying susceptibility and resistance to therapies.
Dr. Varmus’ team acknowledges that their model lacks the paracrine effects that are typically required for tumor growth in complex organisms. However, this model is based on human cells rather than non-human cells used in current SCLC models and allows characterization of a large population of cells which can be manipulated in coordinated fashion. The team also noted that the profiles of single-cell RNA in PNECs are heterogeneous. When the levels of RB are reduced in their PNEC-like cells, resemblance of early-stage SCLC occurs. However, when both RB and TP53 are reduced in these cells, the transcriptome favors cell proliferation and a more advanced SCLC-like tumorigenic behavior.
In this project Dr. Varmus and his team validated the induction of PNECs from hECSs by inhibiting the NOTCH pathway and inactivating RB. These results have been recently published in the Journal of Experimental Medicine (Chen, et al. 2019). Dr. Varmus’ team is continuing this study with support from another funding agency; they are examining the mechanisms by which NOTCH and RB disruptions yield PNEC development and exploring the possibility of studying tumor growth of these cells in other locations, such as in lung tissue. If successful, Dr. Varmus’ team will generate more accurate cell and xenograft mouse models, significantly advancing the understanding of SCLC disease progression while also providing the research community additional tools to help generate more effective treatment for SCLC.
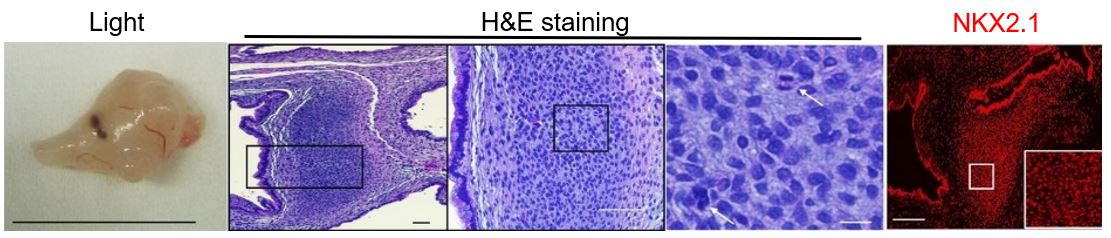 Reducing RB and TP53 in Notch activated PNECs allows xenograft tumor growth resembling SCLC. Left segment depicts a generated tumor under light microscopy; scale bar of 1 cm. Center three segments depict H&E staining of the tumor at increasing magnifications (white arrows point to cells with mitotic figures); scale bars from left to right: 200 µm, 100 µm, 10 µm. Right segment depicts staining of tumor tissue for NKX2.1, a marker of lung tissue; scale bar of 200 µm. Figure adapted from Chen HJ, Poran A, Unni AM, et al. 2019. Generation of pulmonary neuroendocrine cells and SCLC-like tumors from human embryonic stem cells. Journal of Experimental Medicine 216-3:674–687.
Reducing RB and TP53 in Notch activated PNECs allows xenograft tumor growth resembling SCLC. Left segment depicts a generated tumor under light microscopy; scale bar of 1 cm. Center three segments depict H&E staining of the tumor at increasing magnifications (white arrows point to cells with mitotic figures); scale bars from left to right: 200 µm, 100 µm, 10 µm. Right segment depicts staining of tumor tissue for NKX2.1, a marker of lung tissue; scale bar of 200 µm. Figure adapted from Chen HJ, Poran A, Unni AM, et al. 2019. Generation of pulmonary neuroendocrine cells and SCLC-like tumors from human embryonic stem cells. Journal of Experimental Medicine 216-3:674–687.
Publication:
Chen HJ, Poran A, Unni AM, Huang SX, Elemento O, Snoeck HW, and Varmus H. 2019. Generation of pulmonary neuroendocrine cells and sclc-like tumors from human embryonic stem cells. Journal of Experimental Medicine 216-3:674–687.
Link:
Last updated Thursday, November 13, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
