Lung Cancer



Posted November 29, 2017
David B. Shackelford, Ph.D., and Steven M. Dubinett, M.D., Division of Pulmonary and Critical Care Medicine
David Geffen School of Medicine and Jonsson Comprehensive Cancer Center, University of California, Los Angeles

Dr. David Shackelford

Dr. Steven Dubinett
Lung cancer is the leading cause of cancer-related deaths worldwide, and approximately 85% of those cases are classified as non-small cell lung cancer (NSCLC). There are currently no effective strategies for using medication to prevent or lower the risk of lung cancer (i.e., chemoprevention or cancer interception). Moreover, the few targeted therapies available are effective for a minority of NSCLC patients. This lack of preventative and targeted therapies leaves many NSCLC patients with limited therapeutic options, underscoring the need for scientists to find new and effective treatment strategies for NSCLC.
Although the connection between cellular metabolism and tumorigenesis has long been established, understanding the mechanisms that connect the signaling pathways controlling metabolism and cell growth has only recently begun. The growth and reproduction of any cell requires the ability to acquire and metabolize nutrients (glucose and glutamine are the primary energy sources). Growth factors and energy stress produce signals that communicate to the cell whether or not nutrients are sufficient enough to ensure successful cellular proliferation. With support from a fiscal year 2012 (FY12) Lung Cancer Research Program Translational Research Partnership award Drs. Shackelford and Dubinett explored inhibiting tumorigenesis by targeting growth and survival pathways combined with metabolic stress agents in NSCLC bearing mutations in the LKB1, KRAS, and epidermal growth factor receptor (EGFR) genes.
Dr. Shackelford's foundational research showed that inhibition with phenformin - a small molecule drug that induces metabolic stress in tumor cells led to tumor cell apoptosis selectively in LKB1-deficient NSCLC cells. The LKB1 gene is a known tumor suppressor that is inactivated in approximately 20-30% of NSCLC patients. LKB1 regulates energy balance in the cells. Mutations in LKB1 render lung tumor cells unable to cope with phenformin induced energy stress. Even though both human lung cell cultures and mouse models of NSCLC eventually grew resistant to phenformin, these foundational studies also showed that one mechanism of resistance may be through hyper-activation of the mTORC1 protein complex, which regulates glycolysis and actively drives cell growth. Drs. Shackelford and Dubinett strategized to increase the cells' ability to sense metabolic stress by co-targeting tumors using phenformin to induce energetic stress with targeted inhibitors against mTORC1 to suppress glycolysis and phenformin resistance. Their results, reported in Cancer Research (2015), demonstrated that combining MLN0128, an mTOR inhibitor, with phenformin restricted tumor growth and enhanced cell death in mouse models of lung cancer and human lung adenocarcinoma cell cultures.
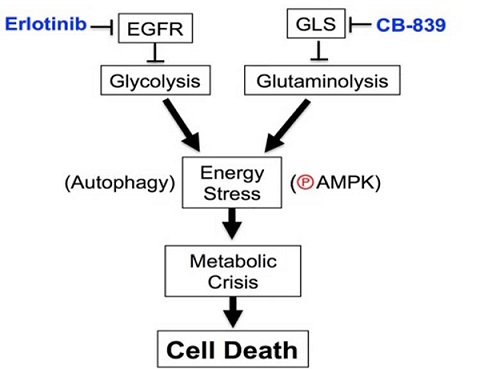
Success with LKB1-deficient NSCLC encouraged the team to study additional energy stress-inducing strategies to mitigate proliferation of NSCLC cells with EGFR mutations, which affects approximately 15% of NSCLC patients in the United States. Glycolysis and glutaminolysis are attractive target pathways for combination therapies because they are key metabolic pathways that ensure EGFR-mutant NSCLC proliferation. Dr. Shackelford's and Dr. Dubinett's team demonstrated that the combined use of the EGFR tyrosine kinase inhibitor, erlotinib, and a glutaminase inhibitor, CB-839 induced rapid tumor cell death and tumor regression in mice xenografts bearing human EGFR-mutant NSCLC. They discovered that co-suppression of glycolysis and glutaminolysis with erlotinib and CB-839 induced energetic stress in EGFR-mutant tumors in vivo; marked by activation of the AMPK pathway, which governs glucose and lipid metabolism, and autophagy ultimately led tumor cell death. These results were published in Cell Reports (2017) and support the rationale to combine erlotinib and CB-839 for treatment of NSCLC patients with EGFR mutations, which is currently being tested in a Phase I clinical trial (NCT02071862, Calithera Biosciences, Inc.).
The ability to reduce lung cancer mortality requires a major shift from focusing research on later-stage disease to early detection and chemoprevention. Dr. Shackelford's and Dr. Dubinett's research has advanced this approach by uncovering factors that are critical to the progression of lung carcinogenesis in mouse and human models. Their work thus far has explored several molecular pathways that control cell growth and metabolism, and these may eventually be exploited to prevent or treat multiple lung tumor subtypes.
Publications:
Momcilovic M, Bailey ST, Lee JT, Fishbein MC, Magyar C, Braas D, Graeber T, Jackson NJ, Czernin J, Emberley E, Gross M, Janes J, Mackinnon A, Pan A, Rodriguez M, Works M, Zhang W, Parlati F, Demo S, Garon E, Krysan K, Walser TC, Dubinett SM, Sadeghi S, Christofk HR, Shackelford DB 2017. Targeted inhibition of EGFR and glutaminase induces metabolic crisis in EGFR mutant lung cancer. Cell Rep 18(3):601-610.
Momcilovic M, McMickle R, Abt E, Seki A, Simko SA, Magyar C, Stout DB, Fishbein MC, Walser TC, Dubinett SM, Shackelford DB. 2015. Heightening energetic stress selectively targets LKB1-deficient non-small cell lung cancers. Cancer Res 75(22):4910-4922.
Link:
Last updated Monday, March 10, 2025














