Kidney Cancer



Posted March 23, 2021
Maria Czyzyk-Krzeska, M.D., Ph.D., University of Cincinnati

Dr. Maria Czyzyk-Krzeska
Clear cell renal cell carcinoma (ccRCC) represents the predominant subtype diagnosed and the most common malignant form of kidney cancer. Despite strong associations established between tobacco smoke (TS) and ccRCC development, there is a gap in understanding the genetic and signaling signatures of TS as a risk factor driving ccRCC growth. This, taken together with the high prevalence of smoking among active military personnel and Veterans, reveals a need for studies to elucidate the molecular mechanisms by which smoking promotes renal cancer. Dr. Maria Czyzyk-Krzeska, with funding from a Fiscal Year 2013 (FY13) Peer Reviewed Cancer Research Program (PRCRP) Idea Award (IA) with Special Focus, investigated TS with the aim of uncovering its downstream effects on tumor pathobiology of ccRCC.
Dr. Czyzyk-Krzeska hypothesized that carcinogens present in TS mediate genetic changes that affect tumor growth. To identify these changes, she and her team conducted multiomics analyses using tissues obtained from renal tumors of male patients who were lifetime smokers (LTS) versus those who were never smokers (NS).
In a study recently published in The Journal of Clinical Investigation, Dr. Czyzyk-Krzeska and her team reported their findings from a landscape analysis of primary tumors and paired normal kidney tissues (NKTs) from LTS and NS White males, together referred to as the Cincinnati Tobacco Smoking Cohort (CTSC). The integrated analysis approach utilized transcriptomics, metabolomics, and metallomics to obtain a comprehensive metabolic profile. The transcriptomics studies showed an upregulation of oxidative phosphorylation (OxPhos) genes in ccRCCs from LTS including: (1) genes for mitochondrial calcium and pyruvate transporters; (2) subunits for mitochondrial respiratory complex I, II, III, V; and (3) malate dehydrogenase 1 (MDH1), required for the activity of the malate-aspartate shuttle (MAS), which regulates cytosolic and mitochondrial NAD/NADH ratio. Metabolomics assessment showed a shift away from glycolysis towards OxPhos in generation of adenosine triphosphate (ATP) in ccRCCs from LTS. The alternations included reprogramming of MAS and of the amino acids metabolism, in particular of aspartate (Asp), glutamate (Glu), glutamine (Gln) and histidine (His). To investigate the involvement of metals in the biochemical pathways, the study team conducted metallomic analysis, which displayed higher accumulation and intracellular redistribution of cadmium (Cd) and inorganic arsenic (As) in the NKTs and ccRCCs from LTS, and increased amounts of copper (Cu) in ccRCCs from LTS. Furthermore, Dr. Czyzyk-Krzeska and her team observed relocation of Cu to the mitochondrial cytochrome c oxidase complex IV, an independent validation for the increased activity of the oxidative phosphorylation in ccRCCs from LTS. The model presented in Figure 1 illustrates the TS-dependent metabolic reprogramming of the TCA cycle in ccRCC tumor subtype.
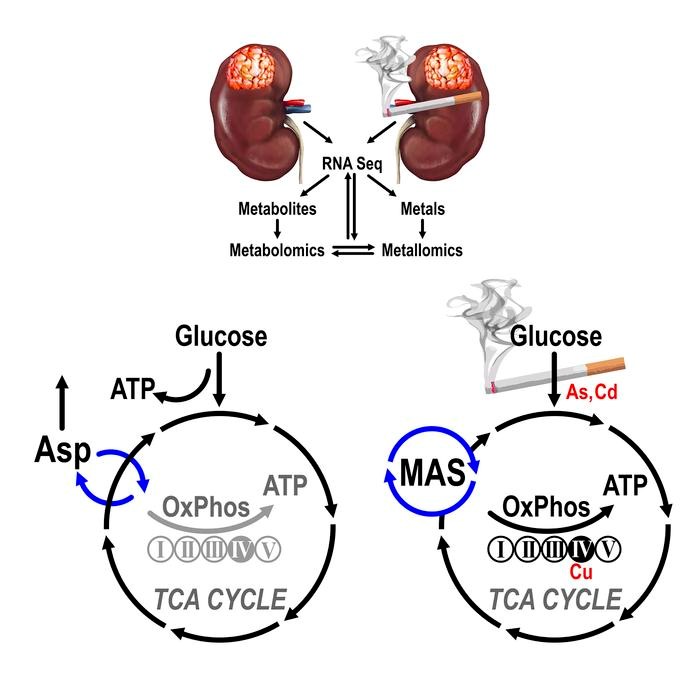
Figure 1. Graphical Abstract: Tobacco smoking induces
metabolic reprogramming of ccRCC.[1]
This unique profile not only provides a better understanding of the disease, but also a novel signaling pathway for development of diagnostic and prognostic biomarkers. Additionally, Dr. Czyzyk-Krzeska, in her publication, alluded to growing interest in OxPhos inhibitory agents, some currently being tested in clinical trials, as potential treatment options for cancers, making the elucidation of this metabolic reprogramming extremely timely. Dr. Czyzyk-Krzeska’s contributions in the field of ccRCC provide a basis for advancement of approaches in the diagnosis, prognosis, and treatment of this disease, with particular impact among the smoking military and civilian patient population. The role of metals in ccRCC pathobiology draws attention to the effects of metals from e-cigarettes and other exposures relevant for military personnel.
Publication:
Reigle J, Secic D, Biesiada J, et al. 2021. Tobacco smoking induces metabolic reprogramming of renal cell carcinoma. The Journal of Clinical Investigation 131(1):e140522. https://doi.org/10.1172/JCI140522.Link:
Public and Technical Abstracts: Effects of Tobacco Smoke (TS) on Growth of Clear Cell Renal Cell Carcinoma (ccRCC)[1] Reprinted from “Tobacco smoking induces metabolic reprogramming of renal cell carcinoma,” by Reigle J, Secic D, Biesiada J, et al. 2021. The Journal of Clinical Investigation 131(1):e140522. Copyright 2020 by The Journal of Clinical Investigation. Reprinted with permission.
Last updated Wednesday, November 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
