Military Operational Medicine (JPC-5)
Novel Stem Cell Model Reveals How Neurons Respond to Stress in PTSD



Posted June 27, 2023
Rachel Yehuda, Ph.D., Icahn School of Medicine at Mount Sinai
This project is managed by the CDMRP under the Psychological Health and Traumatic Brain Injury Research Program on behalf of the Military Operational Medicine Research Program.
 Dr. Rachel Yehuda
Dr. Rachel Yehuda (Photo Provided)
Following a traumatic event, some individuals develop posttraumatic stress disorder (PTSD), a condition that can cause intrusive thoughts, avoidance, and altered mood, among other symptoms that impact daily life.1 Research indicates that genetics may play a role in the development of PTSD and could explain why this disorder does not arise in all individuals exposed to a traumatic event. Understanding the interaction of genes and environment would aid in the diagnosis, prognosis, and therapeutics of PTSD, but the lack of an adequate model that accurately represents the human condition makes this difficult.
To advance the understanding of PTSD, it is necessary to determine the association of blood biomarkers of PTSD and the function of the central nervous system (CNS). With an award from the fiscal year 2014 Broad Agency Announcement for Extramural Medical Research, Dr. Rachel Yehuda and her team utilized advancements in stem cell technology to develop a model for PTSD that allows for study of human neurons. Through comparison of this model with cultured blood cells, the team seeks to improve the understanding of blood biomarkers, mechanisms associated with PTSD, and potential treatment options.
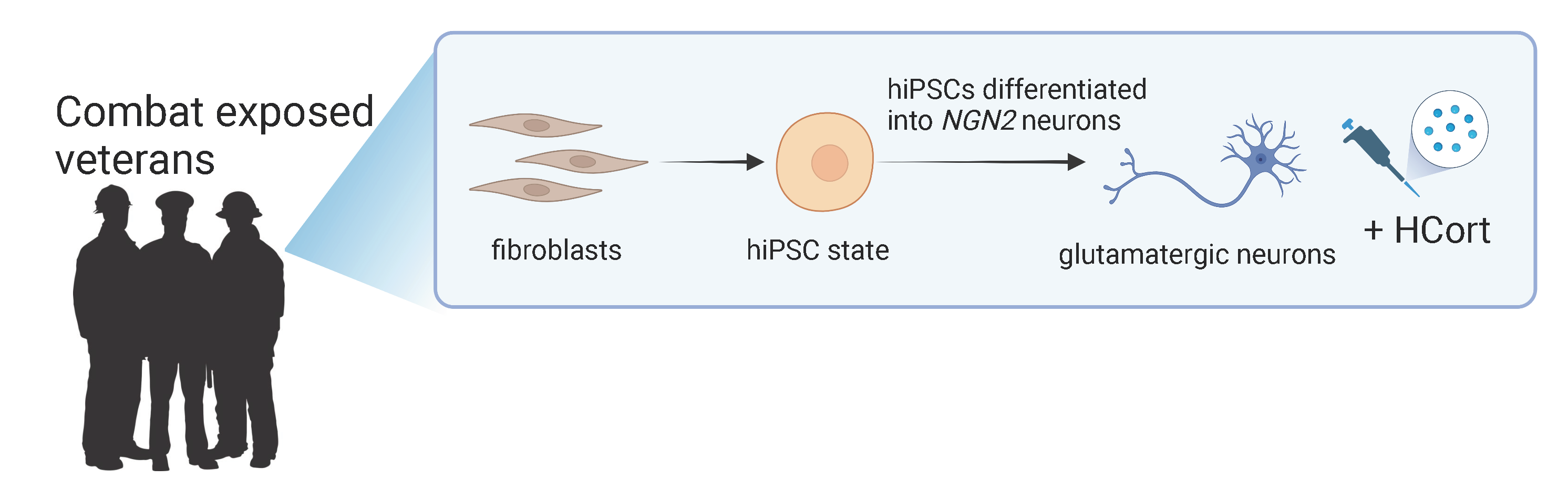 Figure 1: Development of the iNeuron Model. Skin cells (fibroblasts) obtained from combat-exposed veterans with and without PTSD were re-programmed into human induced pluripotent stem cells (hiPSCs) and transformed into excitatory (glutamatergic) iNeurons. To mimic the biological response that occurs in response to trauma, iNeurons were exposed to the stress hormone hydrocortisone (HCort) to reveal unique features of PTSD. (Figure Provided)
Figure 1: Development of the iNeuron Model. Skin cells (fibroblasts) obtained from combat-exposed veterans with and without PTSD were re-programmed into human induced pluripotent stem cells (hiPSCs) and transformed into excitatory (glutamatergic) iNeurons. To mimic the biological response that occurs in response to trauma, iNeurons were exposed to the stress hormone hydrocortisone (HCort) to reveal unique features of PTSD. (Figure Provided)
To develop the model, the team utilized skin samples from combat-exposed Veterans with and without PTSD (Refer to Figure 1). These cells were transformed into stem cells, essentially "resetting” the gene expression to a pre-trauma state. The stem cells were used to generate "iNeurons,” a model featuring neuronal cells that send excitatory signals and have been associated with PTSD. To evaluate underlying genetic differences that may predispose an individual to PTSD, iNeurons from combat Veterans with and without PTSD were exposed to the synthetic version of cortisol, a stress hormone released during traumatic events. Through evaluation of gene expression, the team found that iNeurons from individuals with PTSD had increased sensitivity to cortisol and identified genes that responded differently when exposed to the hormone. These findings could be useful for identifying those at increased risk for PTSD and offer potential therapeutic targets.
Dr. Yehuda and her team plan to continue this work by examining other brain cell types and to further investigate mechanisms involved in the gene/stress interaction and the impact on cell function and symptoms. This work has the potential to lead to significant advances for diagnostic, prognostic, and therapeutic approaches for those living with or at risk for PTSD.
Reference:
1Taylor-Desir M. 2022. What is posttraumatic stress disorder (PTSD)? American Psychiatric Association. Information sheet. https://www.psychiatry.org/patients-families/ptsd/what-is-ptsd
Publication:
Seah C, Breen MS, Rusielewicz T, et al. 2022. Modeling gene × environment interactions in PTSD using human neurons reveals diagnosis-specific glucocorticoid-induced gene expression. Nature Neuroscience 25(11):1434-1445. https://doi.org/10.1038/s41593-022-01161-y
Link:
Last updated Friday, March 7, 2025














