Military Operational Medicine (JPC-5)
Evaluating Sex Differences as a Predictor of Risk for Opioid Use Disorder



Posted November 24, 2020
Elena Chartoff, Ph.D., McLean Hospital
 Dr. Elena Chartoff
Dr. Elena Chartoff
In 2017, the Department of Health and Human Services declared a nationwide public health emergency regarding opioid misuse and, according to the CDC, as many as one in four people prescribed opioids for long-term therapy can struggle with addiction and misuse. As Soldiers return to duty after injury, many are often prescribed opioids for pain management. However, long-term opioid treatment can lead to dependence and result in withdrawal symptoms and eventual self-administration of opioids leading to opioid use disorder (OUD). Preclinical studies for predicting and mitigating opioid misuse are limited, and differences between men and women are not thoroughly understood. In response to a fiscal year 2017 Broad Agency Announcement for Extramural Medical Research, Dr. Elena Chartoff proposed to investigate sex-dependent implications for treating pain and subsequent OUD in an opioid dependence and withdrawal rodent model.
The main objective of this work was to investigate distress intolerance (DI) in male and female rats after an escalating-dose regimen of morphine, followed by abstinence to induce withdrawal. A catheter was implanted to enable oxycodone self-administration to determine addictive behavior. Interestingly, male rats with prior morphine exposure and withdrawal were more likely to self-administer oxycodone. DI was measured using tail flick (TF) latency as a measure of pain intolerance and acoustic startle (AS) amplitude as a measure of startle reactivity. Dr. Chartoff’s team found significant sex-dependent correlations between these measures of DI and oxycodone self-administration. In males, TF withdrawal latency predicted the amount of self-administered oxycodone – the lower the TF latency, the more oxycodone was self-administered. In females, AS amplitude was the more significant indicator of addictive behavior. This supported the hypothesis that pre-opioid exposure baseline DI can be used to predict vulnerability to OUD.
The secondary objective of this project was to test a corticotropin-releasing factor (CRF) receptor type 1 agonist called antalarmin to mitigate morphine withdrawal symptoms, thereby reducing the likelihood of oxycodone self-administration. By blocking corticotropin, a stress hormone known to mediate drug withdrawal, the research team hypothesized that antalarmin would reduce withdrawal symptoms, particularly in females. Preliminary results showed that antalarmin blocked oxycodone self-administration in female rats, but had no effect in males. Although more in-depth studies are needed, the initial results show that antalarmin could prevent withdrawal-induced effects and progression to OUD. Dr. Chartoff and her team will continue to expand these studies by investigating whether antalarmin also interferes with subsequent increased oxycodone use and whether the drug can reduce withdrawal symptoms.
The data from this study suggest that sex differences can predict vulnerability to OUD in patients taking prescription opioids. In this case, male rodents were more susceptible to OUD after being treated with a prescription regimen than females. These results can help inform military physicians when prescribing opioids for pain management, especially regarding those at greater risk for developing OUD. These findings also demonstrate for the first time that DI can predict vulnerability to OUD. This is especially important for military physicians who can incorporate DI as a baseline measure to indicate whether a Service member may be at risk for developing OUD before painkillers are prescribed. This work underscores the need for clinical studies investigating sex differences in addictive behavior and can be used to inform strategies for mitigating the risk of OUD and reducing overall rates of opioid misuse.
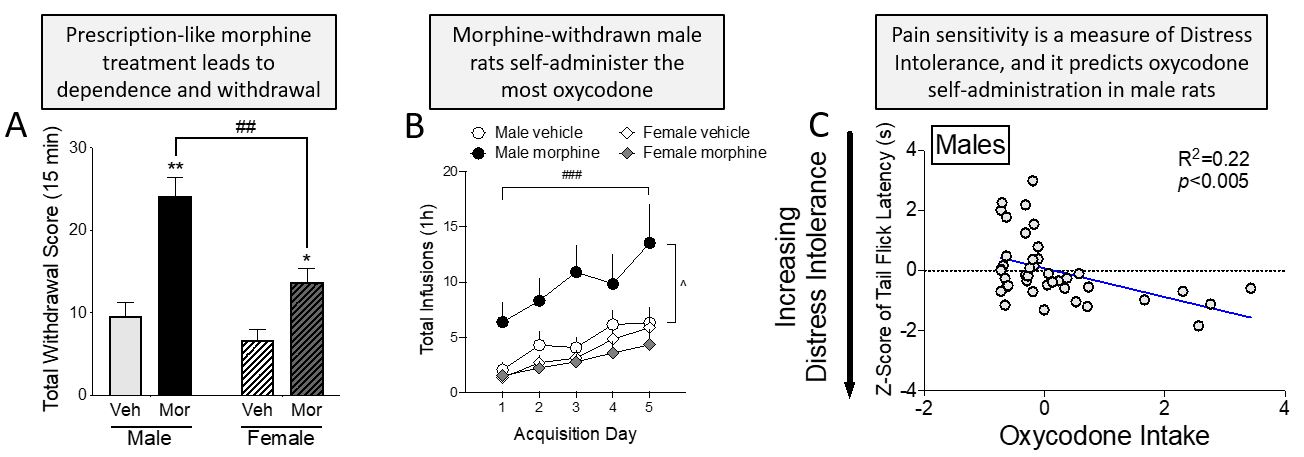 Male and female rats were treated with a prescription-like regimen of morphine for 12 days, which produced robust opioid withdrawal signs upon cessation of drug treatments. Both males and females showed withdrawal signs, but males expressed significantly more than females (A). After a 14-day abstinence period, during which Distress Intolerance (DI) measures such as the warm water Tail Flick Withdrawal test were assessed, rats were implanted with jugular catheters and allowed to self-administer the prescription opioid oxycodone. Under these conditions, rats have control of when they take the drug and how much drug they take. Male rats that had previously been exposed to morphine and morphine withdrawal self-administered the most oxycodone over the initial 5 acquisition days (B). DI measures correlated with the amount of oxycodone self-administered. In (C) we show an example of how the faster a male, but not a female, rat withdraws its tail from the warm water (indicative of increased Distress Intolerance), the more oxycodone it will eventually self-administer.
Male and female rats were treated with a prescription-like regimen of morphine for 12 days, which produced robust opioid withdrawal signs upon cessation of drug treatments. Both males and females showed withdrawal signs, but males expressed significantly more than females (A). After a 14-day abstinence period, during which Distress Intolerance (DI) measures such as the warm water Tail Flick Withdrawal test were assessed, rats were implanted with jugular catheters and allowed to self-administer the prescription opioid oxycodone. Under these conditions, rats have control of when they take the drug and how much drug they take. Male rats that had previously been exposed to morphine and morphine withdrawal self-administered the most oxycodone over the initial 5 acquisition days (B). DI measures correlated with the amount of oxycodone self-administered. In (C) we show an example of how the faster a male, but not a female, rat withdraws its tail from the warm water (indicative of increased Distress Intolerance), the more oxycodone it will eventually self-administer.
Link:
Public and Technical Abstracts: Sex Differences in the Ability to Predict and Treat Opiate Abuse
Last updated Thursday, December 5, 2024














