Duchenne Muscular Dystrophy
Steps Forward in Treating Duchenne Muscular Dystrophy with Gene Repair Therapy



Posted September 4, 2020
Dongsheng Duan, Ph.D., University of Missouri
Charles Gersbach, Ph.D., Duke University

Dr. Dongsheng Duan,
University of Missouri

Dr. Charles Gersbach,
Duke University
Duchenne muscular dystrophy (DMD) is a disorder caused by a mutation of the dystrophin gene, which causes the heart and skeletal muscle cells to become more susceptible to cellular injury. Death from heart-based complications (cardiomyopathy) occurs in 40% of individuals with DMD. Current treatments for Duchenne cardiomyopathy are to use symptom-relieving and/or cardio-protective medications that only delay disease progression. None of these therapies address the underlying genetic defect.
With funding from a Duchenne Muscular Dystrophy Research Program Fiscal Year 2016 (FY16) Investigator-Initiated Research Award (IIRA), Drs. Duan and Gersbach evaluated a gene therapy approach to treat DMD-associated cardiomyopathy. The research team used gene editing with clustered regularly interspaced palindromic repeat (CRISPR)/CRISPR-associated endonuclease 9 (Cas9) with adeno-associated virus (AAV) as the delivery system to target muscle cells in the DMD mdx mouse model. Initial studies demonstrated widespread dystrophin restoration in the heart; however, dystrophin levels were too low to restore hemodynamic function and no dystrophin expression was detected in skeletal muscles. To address this issue a new strategy was pursued where increased levels of guide RNA (gRNA) were used to help Cas9 find the correct gene target. Cardiomyopathy studies with increased gRNA were performed in female mdx mice because they present more severe cardiac disease than males. Conversely, the same experiments were done using male mdx mice to target skeletal muscle. The increase of gRNA improved CRISPR editing and dystrophin restoration in both female (heart) and male (skeletal muscle) cases. Importantly, this optimized CRISPR therapy significantly reduced fibrosis in both heart and skeletal muscle, enhanced skeletal muscle contractility, and cardiac blood pump function for 18 months. These results suggest that by optimizing the gRNA level, this method is a possible approach to achieve long-term, CRISPR-based gene therapy in both skeletal and heart muscle. The research from this FY16 IIRA were published in JCI Insight, "AAV CRISPR editing rescues cardiac and muscle function for 18 months" .
In a subsequent related study published in Nature Medicine, "Long-Term Evaluation of AAV-CRISPR Genome Editing for Duchenne Muscular Dystrophy," Dr. Gersbach's team along with other collaborators investigated whether the AAV-CRISPR technique was a solution for muscle cells lacking dystrophin. During the course of their experiments, the research team examined different types of AAV for delivery of CRISPR-Cas9 treatment and compared IM versus IV injection delivery in newborn and adult mdx mice. Key findings from this study regarding AAV delivery and age included: (1) mice injected IM as adults had a significant decrease in gene editing levels over time, (2) dystrophin protein was restored (and detected) from IV-treated mice for at least 1 year after a single administration, and (3) genes in non-targeted tissues were also edited at marginal levels. There was also a substantial finding related to the immune response from the use of Cas9, which is derived from bacteria. A significant host immune response was avoided if AAV-CRISPR/Cas9 was administered to newborn mice as compared to adults. The researchers concluded that despite the presence of a host response to Cas9 and unintended gene modifications, AAV-CRISPR was well-tolerated and resulted in restoration of dystrophin expression for one year with no sign of toxicity.
In a separate study featured on the cover of the journal Molecular Therapy, "AAV9 edits muscle stem cells in normal and dystrophic adult mice", the team further explored the possibility of correcting dystrophin gene mutation in muscle stem cells. Correcting the mutation in muscle stem cells is important because muscle cells regenerated from the edited stem cells will no longer carry the mutation. Hence, a one-time therapy would result in continuous dystrophin expression in regenerated muscle cells. In this study, the team showed that intravenous delivery of the CRISPR editing machinery with an AAV9 vector can edit dystrophic muscle stem cells. Further, the team showed that new muscle regenerated from the edited stem cells expressed dystrophin. These findings suggest that AAV9 CRISPR gene editing therapy may potentially provide a life-long correction of the genetic mutation in DMD and other muscle diseases. One-time treatment may provide a source of gene-edited cells throughout the life.
Drs. Duan and Gersbach and their research teams have paved the way toward DMD gene therapy treatment by demonstrating that CRISPR-Cas9 gene editing works in mdx mice. While more research is needed to evaluate CRISPR-Cas9's efficiency, possible immune responses, and potential toxicity in larger DMD animal models, there is potential for this therapy to be translated to patients with DMD. Such an approach would potentially transform the lives of those who have DMD and their families.
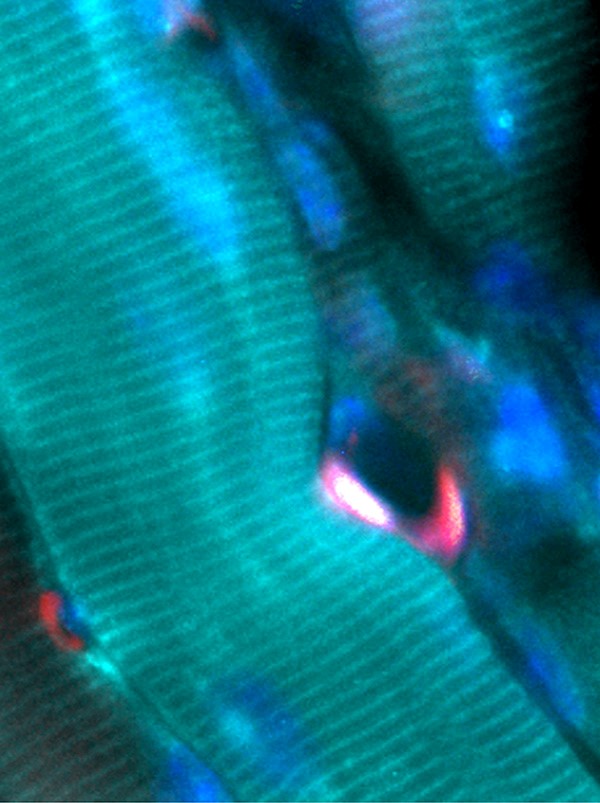
AAV9 CRISPR edits muscle stem cells in dystrophic muscle. Red, muscle stem cells; Blue, myonuclei; Emerald green, myofiber. Image taken by Dr. Michael Nance (University of Missouri).
Publications:
Hakim CH, Wasala NB, Nelson CE, et al. 2018. AAV CRISPR editing rescues cardiac and muscle function for 18 months. JCI Insight 3(23): e124297
Nelson CE, Wu Y, Gemberling MP, et al. 2019. Long-term evaluation of AAV-CRISPR genome editing for Duchenne muscular dystrophy. Nat Med 25(3):427-432. doi:10.1038/s41591-019-0344-3.
Nance ME, Shi R, Hakim CH, et al. 2019. AAV9 edits muscle stem cells in normal and dystrophic adult mice. Molecular Therapy 27(9):1568-1585
Link:
Public and Technical Abstracts: Treating Duchenne Cardiomyopathy in the Mouse Model by Gene Repair
Publications:
Hakim CH, Wasala NB, Nelson CE, et al. 2018. AAV CRISPR editing rescues cardiac and muscle function for 18 months. JCI Insight 3(23): e124297
Nelson CE, Wu Y, Gemberling MP, et al. 2019. Long-term evaluation of AAV-CRISPR genome editing for Duchenne muscular dystrophy. Nat Med 25(3):427-432. doi:10.1038/s41591-019-0344-3.
Nance ME, Shi R, Hakim CH, et al. 2019. AAV9 edits muscle stem cells in normal and dystrophic adult mice. Molecular Therapy 27(9):1568-1585
Link:
Public and Technical Abstracts: Treating Duchenne Cardiomyopathy in the Mouse Model by Gene Repair
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
