Combat Readiness-Medical
A Nanotechnology-Enabled Biosynthetic Whole-Blood Surrogate for Hemostatic Resuscitation



Posted March 10, 2023
Michael Bruckman, Ph.D., Haima Therapeutics, LLC

(Photo Provided)
Uncontrolled hemorrhage (bleeding) is recognized as the leading factor in medically preventable death from traumatic injury, especially on the battlefield. Advances in injury management, such as tourniquet use and damage control resuscitation, have largely reduced fatality rates, but significant challenges persist. Damage control resuscitation is a non-surgical intervention that seeks to limit blood loss and prevent the lethal cascade of physiological events. One facet of this approach is the administration of fluids, whole blood, and/or targeted blood products (red blood cells, platelets, plasma), to reduce morbidity and mortality from traumatic hemorrhage. Fresh or cold-stored whole blood is the ideal choice for blood transfusion, but conditions in the battlefield may limit availability for immediate treatment of casualties. Development of a synthetic whole blood substitute could augment efforts to establish in-theater blood banks, significantly reduce mortality associated with hemorrhage and related trauma-induced coagulopathy, and reduce the logistical burden of storage and administration of blood products, especially in scenarios with a limited natural blood component supply or prolonged/remote care.
Under the fiscal year 2019 (FY19) Combat Readiness-Medical Research Program's Rapid Development and Translational Research Award, Dr. Michael Bruckman of Haima Therapeutics, LLC, leads a collaborative team that is developing a dried, whole-blood surrogate that can address logistical challenges related to the availability of whole blood in austere battlefield environments. To pursue this approach, Haima Therapeutics has teamed with biotechnology companies to combine, into a single product, blood component surrogates that are already in development. Combination of the nanoparticle-based, red blood cell surrogate, ErythroMer (Kalocyte, Inc.); platelet substitute, SynthoPlate (Haima Therapeutics); and freeze-dried plasma (FDP) are anticipated to serve as a biosynthetic whole-blood surrogate for remote damage control and resuscitation. To create this novel agent, the study team must establish the compatibility of the individual components when mixed with FDP and test the clotting and oxygenation function of the co-formulation. The final product will be evaluated in an in vivo model of acute massive hemorrhage and resuscitation.
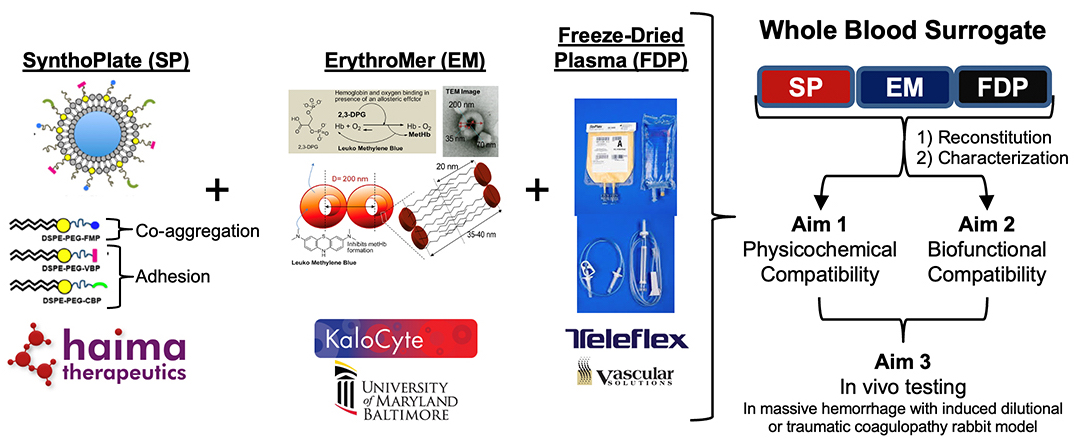
This award's achievements will also significantly impact other Department of Defense-funded research on blood products for resuscitation, including the Defense Advanced Research Projects Agency's (DARPA) Fieldable Solutions for Hemorrhage with bio-Artificial Resuscitation Products (FSHARP) program. If successful, this work may lead to U.S. Food and Drug Administration-regulated clinical trials. The availability of a synthetic whole-blood substitute would significantly enhance care capabilities in pre-hospital environments and reduce preventable deaths from battlefield injuries.
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
