


Posted July 3, 2013
Carla Hand, Ph.D., University of North Carolina, Chapel Hill
 Bone marrow failure can be caused by whole body exposure to even low doses of radiation. Simple, inexpensive, and easily accessible therapies for bone marrow failure due to toxic radiation exposure are currently lacking. Possible terrorist scenarios involving the use of radioactive materials include dispersal of radioactive substances with or without the use of conventional explosives, attacks on nuclear reactors, as well as the detonation of nuclear weapons. The terrorist attacks on the United States on September 11, 2001 raised awareness that the United States is, in fact, vulnerable.
Bone marrow failure can be caused by whole body exposure to even low doses of radiation. Simple, inexpensive, and easily accessible therapies for bone marrow failure due to toxic radiation exposure are currently lacking. Possible terrorist scenarios involving the use of radioactive materials include dispersal of radioactive substances with or without the use of conventional explosives, attacks on nuclear reactors, as well as the detonation of nuclear weapons. The terrorist attacks on the United States on September 11, 2001 raised awareness that the United States is, in fact, vulnerable.
In Dr. Carla Hand's Bone Marrow Failure Research Program Fiscal Year 2010 Exploration - Hypothesis Development Award, she demonstrated that a novel erythropoietin-mimetic peptide has potential as a treatment for bone marrow failure in the murine model. The researchers have shown that the erythropoietin-mimetic peptide improves survival in mice that have been exposed to ionizing radiation using whole body exposure (Figure 1). Additionally, the experiments revealed that a single dose of the erythropoietin-mimetic peptide inhibits systemic tumor necrosis factor (TNF) production in mice exposed to radiation (Figure 2). These remarkable results showed that erythropoietin-mimetic peptide could turn off the inflammatory process following radiation. Currently, the molecular mechanism of this protection is under study.
The discovery and development of a post-radiation exposure treatment for bone marrow failure is essential for the protection of both citizens and members of the armed forces in case of the detonation of a nuclear weapon, or an accident at a nuclear facility. The national importance of these projects cannot be overestimated.
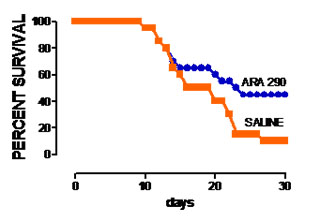
Figure 1: Erythropoietin-mimetic peptide improves survival in a murine model of radiation induced bone marrow failure. Twenty mice per group were exposed to 796 cGy total body radiation. Mice were dosed with erythropoietin-mimetic peptide at 30 µg/kg for 29 days and monitored for 30 days.
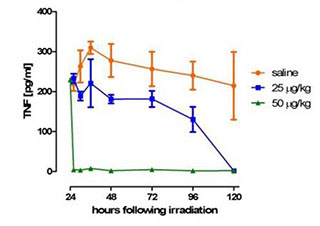
Figure 2: Administration of erythropoietin-mimetic peptide at 24 hours post-radiation exposure inhibits TNF production. Four groups of mice (n=3) were irradiated with 15 Gy partial body irradiation and dosed with erythropoietin-mimetic peptide at the indicated concentration at 24 hours after irradiation. Serum samples were collected at 2 hours, 4 hours, 12 hours, 24 hours, 48 hours, 72 hours, and 96 hours after ARA 290 dosing and assayed for TNF-α concentration.














