Breast Cancer
Dual-Loading Antibody-Drug Conjugates for Combating Breast Cancer Tumor Heterogeneity
and Drug Resistance



Posted June 23, 2023
Kyoji Tsuchikama, Ph.D., UTHealth Houston
 Dr. Kyoji Tsuchikama
Dr. Kyoji Tsuchikama
(Photo Provided)
Intratumor heterogeneity of human epidermal growth factor receptor 2 (HER2) expression presents an important challenge for the treatment of breast cancer and is associated with high relapse rates due to acquired treatment resistance, aggressive growth, and poor survival. Antibodies coupled with a payload (cytotoxic agent), known as antibody�drug conjugates (ADCs), have been shown to provide targeted therapy delivery to HER2-positive breast cancer tumors for increased efficacy and decreased adverse effects. However, intratumor HER2 heterogeneity limits the efficacy of ADCs. Recently, the concept of co-delivery of two separate payloads from a single antibody (dual-drug ADC) has been suggested as a potential method to overcome this challenge. With support from a fiscal year 2017 Breast Cancer Research Program Breakthrough
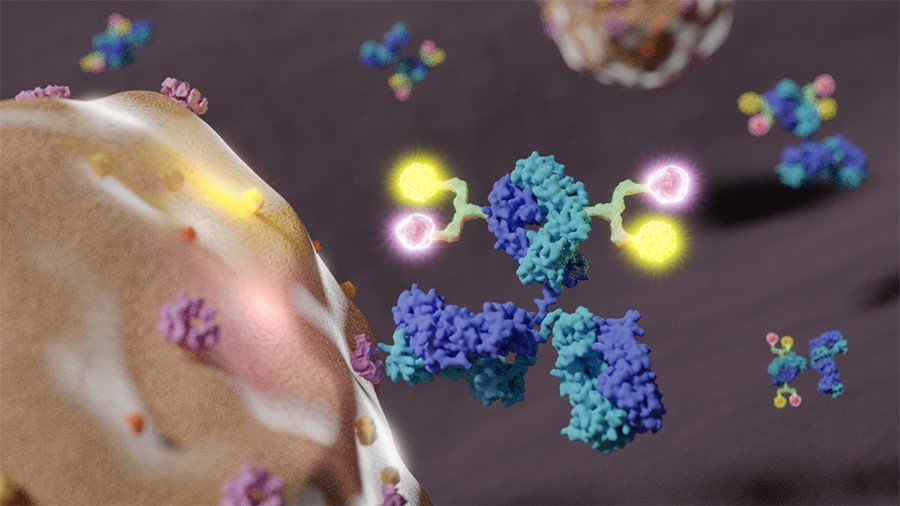 Figure 1: A dual-loading antibody-drug conjugate (ADC) is binding to a receptor on the surface of a breast cancer cell, delivering two distinct antitumor payloads.
Figure 1: A dual-loading antibody-drug conjugate (ADC) is binding to a receptor on the surface of a breast cancer cell, delivering two distinct antitumor payloads.
(Provided by Dr. Kyoji Tsuchikama)
Dr. Tsuchikama and his team reported in Nature Communications their production of both single-drug ADCs, containing payload molecules monomethyl auristatin E or monomethyl auristatin F, and dual-drug ADCs, containing both payloads to kill a wider range of breast cancer cells by combining their different molecular and pharmacological properties. The team created ADCs using a novel glutamic
Overall, Dr. Tsuchikama�s linker technology improved tumor-targeting specificity and payload delivery efficiency of dual-drug ADCs in breast cancer mouse models. These results demonstrate that the therapeutic potential of their dual-drug ADCs is a step toward overcoming the challenges associated with breast cancer heterogeneity and drug resistance. Further assessment of dual-drug ADC toxicity and safety profiles in preclinical models may pave the way for evaluation of dual-drug ADCs in clinical trials for the treatment of refractory HER2-positive breast cancer.
Publication:
Yamazaki CM, Yamaguchi A, Anami Y, et al. 2021. Antibody-drug conjugates with dual payloads for combating breast tumor heterogeneity and drug resistance. Nature Communications 12(1):3528.
Last updated Thursday, November 6, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
