Breast Cancer
Inhibition of Myeloid-Derived Suppressor Cell Biogenesis to Improve
the Efficacy of Immune Checkpoint Inhibitors
in Triple-Negative Breast Cancer



Posted October 27, 2023
Scott I. Abrams, Ph.D. and Michael J. Nemeth, Ph.D., Health Research, Inc., Roswell Park Division
Triple-negative breast cancer (TNBC) is a difficult-to-treat cancer that often spreads faster than other breast cancers and has higher rates of recurrence, leading to poorer outcomes.1 In addition, TNBC cells lack receptors targeted by some breast cancer treatments thereby limiting the treatment options for this aggressive tumor type. Immune checkpoint inhibitors (ICIs) are drugs whose activity does not rely on these absent receptors. Instead, ICIs work by preventing immune checkpoint proteins from suppressing the ability of immune cells to destroy cancer cells thereby boosting the antitumor activity of the immune system. TNBC tumors, however, may negate the effects of ICIs by inducing the development of myeloid-derived suppressor cells (MDSCs). MDSCs are derived from immature bone marrow cells, also known as myeloid progenitors, that would otherwise develop into mature blood cells. Normally, MDSCs exist briefly to turn off the immune response after an acute injury or cellular damage. However, TNBC tumors induce prolonged and persistent accumulation of MDSCs, which leads to continued immune suppression and limits the efficacy of ICIs.
 Dr. Scott I. Abrams
Dr. Scott I. Abrams
(Photo Provided)
 Dr. Michael J. Nemeth
Dr. Michael J. Nemeth
(Photo Provided)
With support from a fiscal year 2020 Breakthrough Award - Funding Level 1 - Partnering PI Option, Drs. Scott Abrams and Michael Nemeth aim to improve the efficacy of ICIs by preventing MDSC development, also known as biogenesis. The team hypothesizes that inhibiting the biogenesis of MDSCs and their replenishment from the bone marrow will be more effective than previously developed strategies that reduced or suppressed existing MDSCs but have yet to translate well to clinical use. The team theorizes that the drug brequinar (BRQ), which is being tested in clinical trials for acute myeloid leukemia - a cancer caused by dysregulation of myeloid cell development - may also inhibit MDSC biogenesis. They previously tested BRQ in preclinical models of breast cancer and found that it improved the antitumor effects of ICIs. To further evaluate this and to determine the effects on MDSC biogenesis, the team tested BRQ in conjunction with ICIs using several models of metastatic TNBC.
 Figure 1: Dr. Scott I. Abrams' and Dr. Michael J. Nemeth's lab team that conducted the research into the inhibition of myeloid-derived suppressor cell biogenesis to improve the efficacy of immune checkpoint inhibitors in models of triple-negative breast cancer.
Figure 1: Dr. Scott I. Abrams' and Dr. Michael J. Nemeth's lab team that conducted the research into the inhibition of myeloid-derived suppressor cell biogenesis to improve the efficacy of immune checkpoint inhibitors in models of triple-negative breast cancer.
The researchers first tested BRQ alone using a bone marrow culture system. In their studies, BRQ treatment significantly reduced the prevalence of MDSCs and, consequently, limited their ability to suppress T cells, which are immune cells that play an important role in antitumor response.
To further assess BRQ's activity, the team tested the agent in mouse models of TNBC. MDSCs recovered from BRQ-treated mice showed signs of greater maturation, supporting the hypothesis that BRQ forces the differentiation of myeloid progenitors. Similar to the bone marrow culture studies, BRQ reduced the expression of several MDSC genes associated with immune suppression. Interestingly, despite its influence on the MDSC response, BRQ alone had minimal effect on reducing tumor growth.
The team next examined whether BRQ would enhance the efficacy of ICI treatment using the same preclinical models of TNBC. They tested BRQ in combination with anti-PD-1, an antibody that targets the immune checkpoint protein PD-1, a negative regulator of T cell activity. Anti-PD-1 antibodies, such as pembrolizumab, are approved by the U.S. Food and Drug Administration for the treatment of TNBC and other cancers, making them ideal candidates to test in conjunction with MDSC-reduction strategies. Neither BRQ nor anti-PD-1 alone exerted substantial antitumor effects in preclinical models. However, when combined, these treatments acted synergistically and significantly reduced the growth of the TNBC tumors. The team also observed synergism between BRQ and another ICI, called anti-CTLA-4, confirming that this strategy can be applied to multiple immune-based therapies.
Through further investigations, the team determined that BRQ (alone or combined with anti-PD-1) reduced the prevalence of metastases in the lung, a common site for metastatic spread of TNBC. Additional experiments demonstrated that BRQ treatment inhibited the ability of myeloid progenitors in the bone marrow to develop into MDSCs.
Therapies for TNBC are limited, driving the need for new options and the improvement of existing treatments. The Abrams and Nemeth team's research demonstrates that the addition of BRQ improves the efficacy of existing ICIs and may translate to effective clinical applications in breast cancer patients, in whom MDSCs may be a significant barrier to therapeutic efficacy.
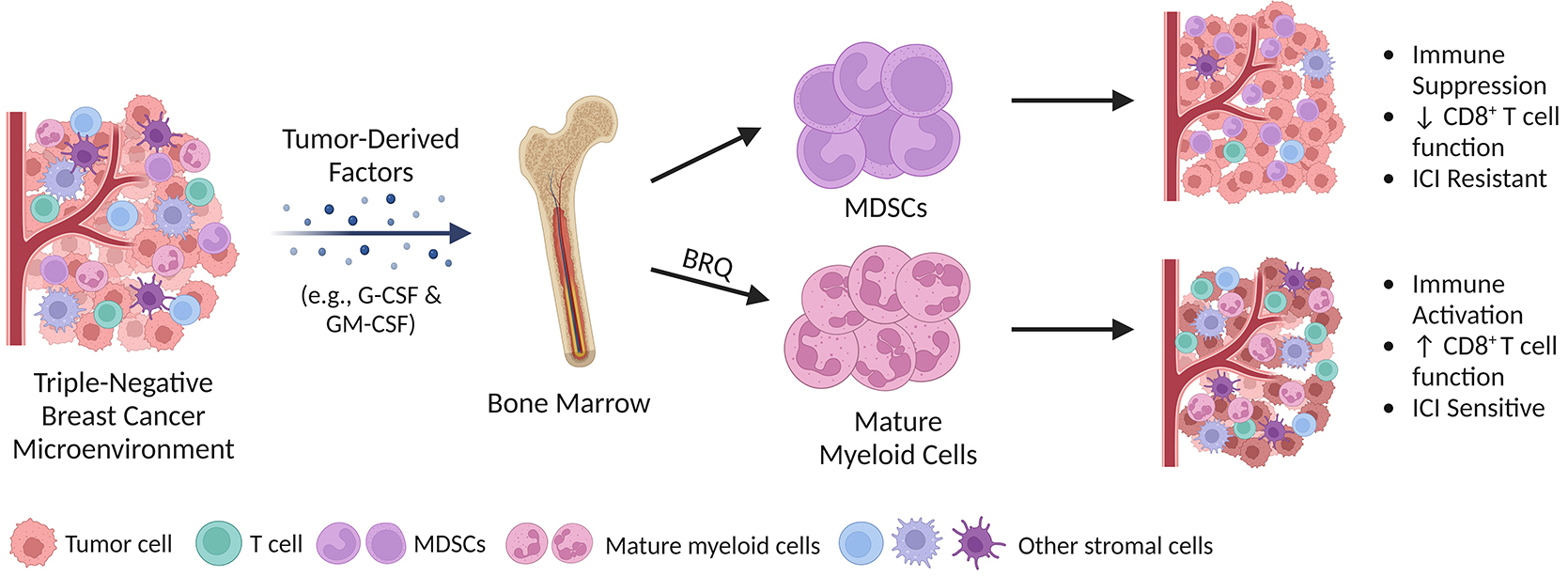 Figure 2: Dr. Scott I. Abrams and Dr. Michael J. Nemeth, from Health Research, Inc., Roswell Park Division, received a fiscal year 2020 Breakthrough Award - Funding Level 1 - Partnering PI Option from the Congressionally Directed Medical Research Programs' Breast Cancer Research Program to study the inhibition of myeloid-derived suppressor cell biogenesis to improve the efficacy of immune checkpoint inhibitors in models of triple-negative breast cancer. The above figure illustrates the impact of the drug brequinar (BRQ) on myeloid-derived suppressor cell (MDSC) biogenesis and immune checkpoint inhibitor therapy. Cancer cells, such as triple-negative breast cancer, secrete soluble factors known as tumor-derived factors (e.g., granulocyte
Figure 2: Dr. Scott I. Abrams and Dr. Michael J. Nemeth, from Health Research, Inc., Roswell Park Division, received a fiscal year 2020 Breakthrough Award - Funding Level 1 - Partnering PI Option from the Congressionally Directed Medical Research Programs' Breast Cancer Research Program to study the inhibition of myeloid-derived suppressor cell biogenesis to improve the efficacy of immune checkpoint inhibitors in models of triple-negative breast cancer. The above figure illustrates the impact of the drug brequinar (BRQ) on myeloid-derived suppressor cell (MDSC) biogenesis and immune checkpoint inhibitor therapy. Cancer cells, such as triple-negative breast cancer, secrete soluble factors known as tumor-derived factors (e.g., granulocyte
References:
1American Cancer Society. Triple-negative Breast Cancer. https://www.cancer.org/cancer/types/breast-cancer/about/types-of-breast-cancer/triple-negative.html
Publications:
Colligan SH, Amitrano AM, Zollo RA, et al. 2022. Inhibiting the biogenesis of myeloid-derived suppressor cells enhances immunotherapy efficacy against mammary tumor progression. The Journal of Clinical Investigation 132(23):e158661. https://doi.org/10.1172/JCI158661
Links:
Last updated Thursday, November 6, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
