Breast Cancer
Predicting Paclitaxel Sensitivity in Breast Cancer Patients



Posted October 18, 2022
Beth Weaver, Ph.D., University of Wisconsin-Madison
Mark Burkard, M.D., Ph.D., University of Wisconsin-Madison
Paclitaxel is frequently used as first-line chemotherapy in breast cancer, but is only effective in about half of patients. Currently, no clinically available biomarker predicts which individuals will respond, causing many patients to receive ineffective treatment and experience treatment-related side effects unnecessarily. With support from a Fiscal Year 2015 Breast Cancer Research Program Breakthrough Award – Funding Level 2 – Partnering Principal Investigator Option, Drs. Beth Weaver and Mark Burkard explored the mechanism by which paclitaxel kills breast cancer cells and the potential of a specific tumor characteristic called chromosomal instability to identify patients that will benefit from paclitaxel treatment.
 Beth Weaver, Ph.D., left, and Mark Burkard, M.D., Ph.D., stand in a laboratory at University of Wisconsin-Madison where they are conducting research aimed at understanding chromosomal instability as a determinant of paclitaxel sensitivity in breast cancer.
(Photo Provided)
Beth Weaver, Ph.D., left, and Mark Burkard, M.D., Ph.D., stand in a laboratory at University of Wisconsin-Madison where they are conducting research aimed at understanding chromosomal instability as a determinant of paclitaxel sensitivity in breast cancer.
(Photo Provided)
Paclitaxel has long been thought to cause cancer cell death by inhibiting the progression of mitosis, which is the process of cell division whereby duplicated chromosomes are evenly separated into two daughter cells. However, findings from research conducted by Drs. Weaver and Burkard and their teams provide key evidence of a mechanism other than mitotic arrest. During cell division, mitotic spindles establish two poles per cell. If there are too many mitotic spindle poles (multipolar), the cell divides its chromosomes incorrectly in more than two directions, resulting in chromosomal instability and many cells that are not viable. As recently reported in the Science Translational Medicine journal, the research teams found that paclitaxel causes multipolar spindles in cancer cells, which leads to chromosome missegregation and, ultimately, cell death, though some breast cancers can evade this drug-induced cell death.
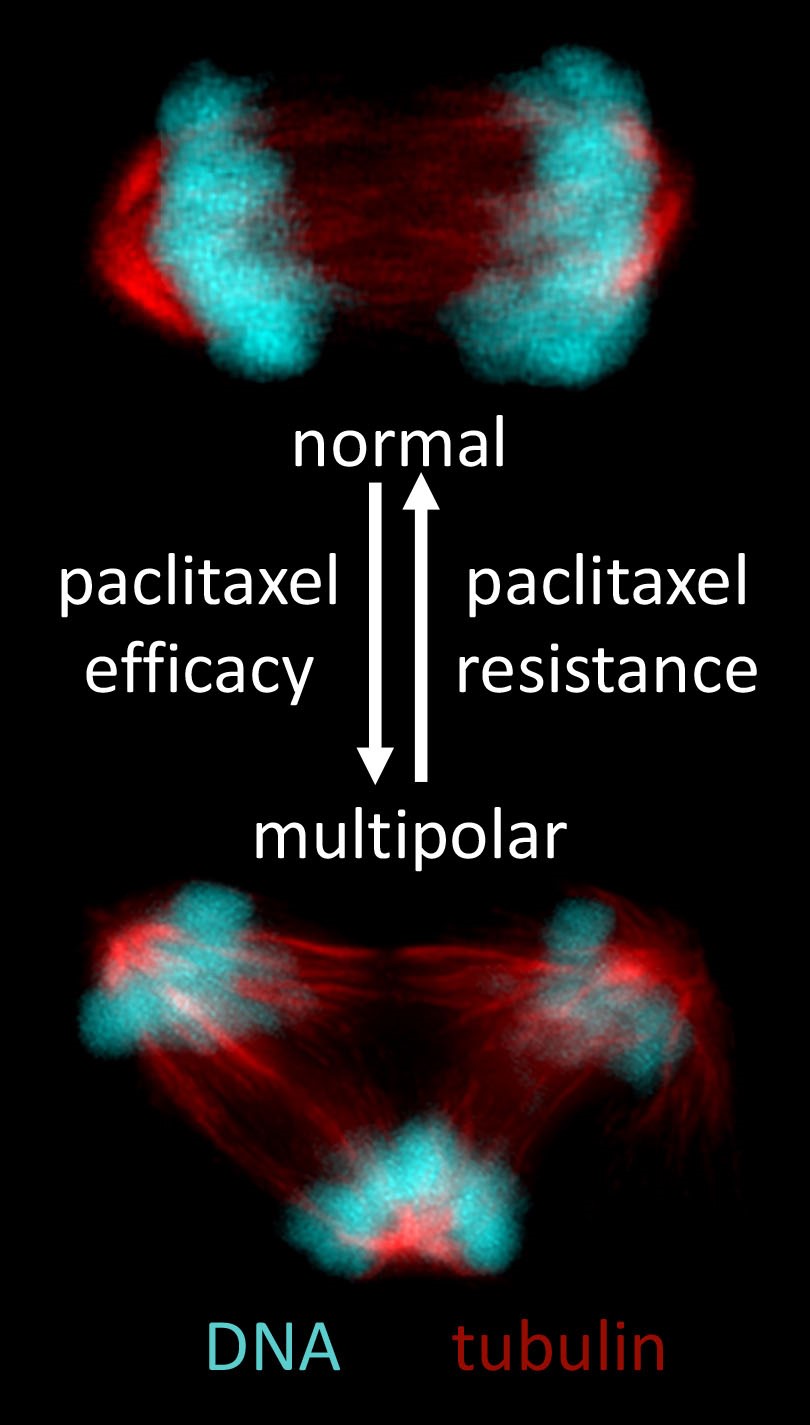 Figure 1: Images of bipolar (top) and multipolar (bottom) anaphase cells (adapted from Scribano CM, et al., Science Translational Medicine journal, 2021). Paclitaxel induces abnormal multipolar spindles in patient tumors. If these persist, it leads to cell death and paclitaxel efficacy. However, focusing of paclitaxel-induced multipolar spindles into near-normal bipolar spindles leads to paclitaxel resistance.
Figure 1: Images of bipolar (top) and multipolar (bottom) anaphase cells (adapted from Scribano CM, et al., Science Translational Medicine journal, 2021). Paclitaxel induces abnormal multipolar spindles in patient tumors. If these persist, it leads to cell death and paclitaxel efficacy. However, focusing of paclitaxel-induced multipolar spindles into near-normal bipolar spindles leads to paclitaxel resistance.
To investigate the mechanism by which paclitaxel kills cancer cells, the teams examined tumor biopsies from patients with primary breast cancer before and after treatment with standard-of-care doses of paclitaxel. Before treatment, most dividing cells displayed normal bipolar mitotic spindles. After treatment, there was a substantial increase in multipolar cells, but no evidence for mitotic arrest. Multipolarity alone, however, did not predict whether a patient would respond to paclitaxel, suggesting there are other factors that contribute to paclitaxel sensitivity.
Multipolar cancer cells can evade cell death by clustering their spindle poles to form near-normal spindles; this spindle focusing reduces chromosome missegregation rates and increases cell viability. The teams explored whether spindle focusing may decrease the sensitivity of cancer cells to paclitaxel treatment. Because spindle focusing cannot be easily assessed in patient tumor tissue, the teams followed the progression of mitosis in multiple human triple-negative breast cancer cell lines exposed to clinically relevant concentrations of paclitaxel. Interestingly, some cell lines readily focused paclitaxel-induced multipolar spindles, while others did not. Rarely did paclitaxel treatment result in mitotic arrest in any of the breast cancer cells. Importantly, after paclitaxel treatment, cells that focused their multipolar spindles had a lower rate of cell death compared to cells that did not focus their multipolar spindles. Further, when the teams increased multipolar divisions in cells, they observed increased sensitivity to paclitaxel. Similarly, when they reduced multipolar divisions, the ability of paclitaxel to cause cell death was decreased. These trends were also seen in mouse models in which tumors initiated with breast cancer cell lines with decreased multipolar divisions were resistant to paclitaxel.
An increase in multipolar divisions leads to chromosome instability. Since chromosome instability can be readily assessed in patient tumor tissue, the researchers investigated whether it had potential as a predictive biomarker of treatment response. Consistent with the observation that increasing chromosomal instability in breast cancer cell lines increased sensitivity to paclitaxel, the teams noted a direct correlation between pretreatment chromosomal instability and paclitaxel response in archived tissue from metastatic breast cancer patients. Breast cancers with higher rates of chromosomal instability before therapy responded preferentially to paclitaxel treatment.
Through their work, Drs. Weaver and Burkard and their teams have confirmed multipolarity and chromosome instability are necessary for the response to paclitaxel. Tumor cells exhibit varying capacities to focus paclitaxel-induced multipolar spindles, which leads to varying levels of chromosome instability and response. As such, treatments that prevent cells from focusing multipolar spindles into bipolar spindles, when administered during paclitaxel therapy, may improve paclitaxel efficacy. Future studies to confirm chromosomal instability as a predictive biomarker for patient response to paclitaxel are important, since such a biomarker would substantially improve patient outcomes by sparing nonresponders the toxicity of paclitaxel and reducing delays in receiving effective treatment.
Publications:
Scribano CM, Wan J, Esbona K, et al. 2021. Chromosomal instability sensitizes patient breast tumors to multipolar divisions induced by paclitaxel. Science Translational Medicine 13(610):eabd4811.
Links:
Chromosomal Instability as a Determinant of Paclitaxel Sensitivity in Breast Cancer
Last updated Thursday, November 6, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
