Breast Cancer
Targeting Senescence May Prevent Chemotherapy-Induced Bone Loss in Breast Cancer Patients



Posted October 01, 2021
Sheila Stewart, Ph.D., Washington University in St. Louis, School of Medicine
 Dr. Sheila Stewart
Dr. Sheila Stewart
Chemotherapy-induced bone loss is a significant problem that negatively impacts the quality of life in breast cancer survivors. While this bone loss has long been attributed to the drop in estrogen that occurs during chemotherapy, similar to that which occurs during menopause, there is substantial data suggesting an estrogen-independent mechanism as well. With support from a Breast Cancer Research Program Breakthrough Award - Funding Level 2, Dr. Sheila Stewart investigated how senescence contributes to bone loss following chemotherapy. Dr. Stewart provides evidence, published in Cancer Research, that senescence drives chemotherapy-induced bone loss and proposes that inhibitors of senescence may reduce bone loss and increase the quality of life for breast cancer survivors.
Although estrogen is known to play a critical role in bone homeostasis, the extent to which chemotherapy-induced bone loss results from decreased estrogen is controversial, and data have suggested that direct effects of chemotherapy drugs on bone may also contribute to this debilitating side effect. To investigate this, Dr. Stewart and team first compared the bone loss initiated by estrogen deprivation to chemotherapy-induced bone loss. The team established a mouse model of chemotherapy-induced bone loss by employing doxorubicin (DOXO), a chemotherapeutic agent used to treat breast cancer patients that has adverse effects on bone. The DOXO mouse model was compared to an ovariectomized (OVX) mouse model of bone loss, where the loss of bone is completely dependent on estrogen deprivation resulting from the removal of the ovaries. In these models, decreased bone volume was greater in DOXO-treated mice relative to OVX mice, illustrating that chemotherapy-induced bone loss was more severe than that due to estrogen loss alone. In a mouse model of postmenopausal chemotherapy treatment, the researchers also found that effects on bone were greater in DOXO-treated mice relative to controls, demonstrating that chemotherapy induced additional bone loss in a low-estrogen setting. Finally, the team demonstrated radiation therapy-induced bone loss in an estrogen-sufficient setting using another mouse model. Collectively, these results support the existence of an estrogen-independent mechanism of chemotherapy- and radiation-induced bone loss.
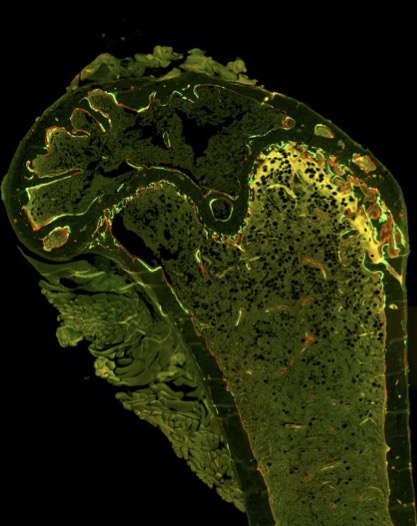 A mouse femur showing calcein (green) and alizarin 7 (red) staining, which were used to indicate regions of bone growth in the mouse models.
A mouse femur showing calcein (green) and alizarin 7 (red) staining, which were used to indicate regions of bone growth in the mouse models.
Chemotherapy drugs used to treat breast cancer induce cellular senescence, and age-related increases in senescent cells were recently shown to drive age-related bone loss. These findings raise the possibility that senescence might be a contributing factor to chemotherapy-induced bone loss. To assess the presence of senescence in the bone following chemotherapy or radiation, the team assayed for changes in senescence-associated secretory phenotype (SASP) factors that senescent cells express. Bone resident cells from tibias of mice that had been treated with chemotherapy or radiation revealed increases in expression of SASP factors relative to non-treated mice. Importantly, the team did not observe increases in these factors in OVX mice, demonstrating that estrogen loss alone is not sufficient to induce senescence in the bone.
Dr. Stewart and team reasoned that if senescence mediates chemotherapy-induced bone loss, then the clearance of senescent cells would protect against this bone loss. To establish this, the team utilized a mouse model harboring a gene that kills senescent cells upon administration of the drug AP20187 (AP). In this model, mice treated with AP exhibited reduced DOXO-induced senescence in the bones, and had significantly higher bone volume compared with mice receiving DOXO alone. These results indicate that senescence is driving the chemotherapy-induced bone loss. Because the p38MAPK-MK2 pathway regulates the expression of many SASP factors and is activated in the bone following chemotherapy, this pathway provides a possible mechanism for regulating the effects of senescent cells and protecting against chemotherapy-induced bone loss. To evaluate this, p38MAPK and MK2 inhibitors were given to the mice treated with DOXO. The inhibitors were observed to reduce SASP factors and mitigate chemotherapy-induced bone loss (with minimal effects on estrogen levels), suggesting that p38MAPK or MK2 inhibition are viable therapeutic strategies to protect against chemotherapy-induced bone loss.
Dr. Stewart and team have identified a role for senescence in chemotherapy-induced bone loss, and illustrated that targeting the p38MAPK-MK2 pathway may help minimize this negative effect. With a FY20 BCRP Breakthrough Award - Funding Level 3 - Partnering PI Option, Dr. Stewart and Partnering PI Dr. Cynthia Ma plan to test a MK2 inhibitor in combination with chemotherapy in a Phase I/II clinical trial. The trial will evaluate safety, as well as the effectiveness of the test compound in combination with chemotherapy to limit disease progression, along with disease- and therapy-induced bone loss in metastatic breast cancer patients. Dr. Stewart's exciting pre-clinical results suggest the ability to develop strategies that mitigate chemotherapy's detrimental impact on bone loss. Because nearly all breast cancer patients, regardless of subtype, are treated with chemotherapeutic agents, this work could positively impact the quality of life of many breast cancer patients.
 Dr. Sheila Stewart and her research team at the Washington University in St. Louis, School of Medicine
Dr. Sheila Stewart and her research team at the Washington University in St. Louis, School of Medicine
Publication:
Yao Z, Murali B, Ren Q, et al. 2021. Therapy-induced senescence drives bone loss. Cancer Research, 80(5):1171-1182.
Link:
Last updated Thursday, February 19, 2026
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
