Breast Cancer
Characterization of Clustered Circulating Tumor Cells to Eliminate Breast Cancer Metastasis



Posted October 28, 2020
Huiping Liu, MD, PhD, Northwestern University

Dr. Huiping Liu
Circulating tumor cells (CTCs) are cancer cells that disseminated from a solid primary tumor, are detectable in the blood, and may lead to metastases. Recent studies have shown that detection of clustered CTCs, as opposed to single CTCs, in the blood is associated with a worse prognosis. However, the mechanisms by which CTCs form clusters and promote the establishment of metastatic lesions remain unclear. With the support of a Fiscal Year 2015 Breakthrough Award – Funding Level 2 – Partnering PI Option, Dr. Huiping Liu sought to characterize the cellular and molecular properties that enable tumor cluster formation and the role of CTC clusters in breast cancer metastasis.
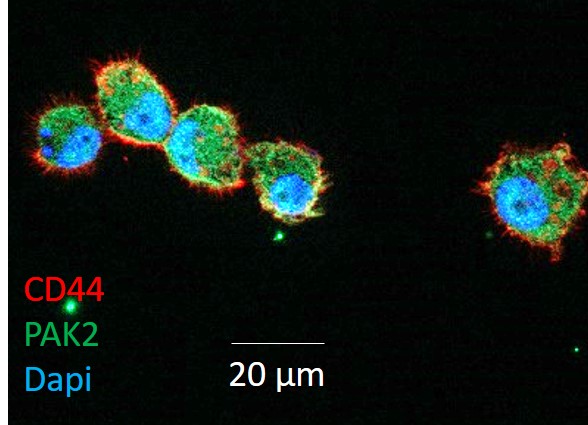
Dr. Liu’s recent publication in Cancer Discovery demonstrated that CTC clusters have more cancer stem cell-like characteristics, which improve their growth, survival, and metastatic potential, compared to single CTCs. Further, she described a specific cell-cell adhesion process by which single CTCs form clusters: CD44 trans-dimerization.

Dr. Liu and her research team employed an established patient-derived xenograft (PDX) lung-metastatic mouse model of breast cancer as a replication of human metastatic disease. They first examined peripheral blood and lung tissue of mice bearing multiple primary tumors derived from implanted mammary tumor cells that were genetically modified to express different (i.e., red or green) fluorescent proteins. Both single-color and dual-color CTC clusters were detectable in blood, as well as in lung metastatic colonies, demonstrating the potential for CTC clusters to form from single CTCs while in circulation, as opposed to pre-formed clusters being released from the tumor. In addition, implantation of CTC clusters in tumor-free mice drastically increased tumorigenesis compared to implantation of single CTCs.
Tumor initiation and recurrence have been linked to the presence of cancer stem cells. CD44 is a protein on the surface of cancer stem cells that plays a role in cell-cell adhesion, cell migration, and invasion from the blood into organ tissues. Comparing single and clustered CTCs from mouse PDX and human tumor samples, the research team observed enriched expression of CD44 in CTC clusters, suggesting that CD44 may play a role in the formation or metastatic potential of CTC clusters. Accordingly, knockdown or deletion of the CD44 gene resulted in reduced clustering, tumor growth, and lung metastases. Importantly, they determined that CTC clustering was dependent on CD44 trans-dimerization (i.e., bonding of CD44 expressed at the interface of two neighboring cells). Analysis of CTCs in blood samples from patients with metastatic breast cancer revealed that longer overall survival correlated with CTCs that did not cluster or express high levels of CD44. These results provided compelling evidence that CTC clusters and CD44 expression have a negative impact on patient outcomes.
Further mechanistic studies by the team identified a region of the CD44 protein sequence that may mediate CD44 trans-dimerization. Interestingly, deletion of that region decreased the formation of CTC clusters to the same effect as treatment with an anti-CD44 antibody. Additional studies examining CD44 dimer-disrupting substitutions, the results of which were recently published in the Journal of Biological Chemistry, may help inform the development of therapeutic strategies to prevent tumor cluster formation and block cluster-mediated metastases.
The team’s characterization of the mechanism by which CD44 contributes to CTC clustering offers new strategies for treatments targeting CD44 and CTCs. Dr. Liu will expand upon these findings, in collaboration with Dr. Alvin Schmaier at Case Western Reserve University, to examine the signaling network associated with CD44, as well as blood cell interactions that contribute to CTC cluster formation. Anti-CD44 therapies have advanced to clinical trials but demonstrate undesirable off-target effects and toxicity. New therapies specifically targeting CD44 trans-dimerization could avoid these toxicities and reduce the metastatic potential of CTCs.
Publications:
Liu X, Taftaf R, Kawaguchi M, et al. 2019. Homophilic CD44 interactions mediate tumor cell aggregation and polyclonal metastasis in patient-derived breast cancer models. Cancer Discovery 9(1):96-113.
Kawaguchi M, Dashzeveg N, Cao Y, et al. 2020. Extracellular domains I and II of cell-surface glycoprotein CD44 mediate its trans-homophilic dimerization and tumor cluster aggregation. Journal of Biological Chemistry 295(9):2640-2649.
Link:
Last updated Thursday, February 19, 2026
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
