Breast Cancer
Enhancing the Cytotoxicity of T-DM1 to Treat HER2+ Breast Cancer



Posted April 7, 2020
Joan Brugge, Ph.D., Harvard Medical School
Jason Zoeller, Ph.D., Harvard Medical School

Overcoming drug resistance is a major hurdle in treating advanced and metastatic breast cancer. Trastuzumab emtansine (T-DM1) is an FDA-approved antibody-drug conjugate (ADC) that is designed to deliver the cytotoxic agent, emtansine, specifically to tumor cells expressing HER2 protein. While the clinical benefit of T-DM1 has been demonstrated in advanced and metastatic HER2+ breast cancer patients, tumors ultimately become resistant, causing patients’ breast cancer to progress to lethal metastatic disease. A family of anti-apoptotic proteins, B-cell lymphoma 2 (BCL-2) and B-cell lymphoma extra-large (BCL-XL), have been found to be upregulated in breast cancers and to contribute to the development of therapy resistance. With the support of a Fiscal Year 2015 Breast Cancer Research Program Breakthrough Award – Funding Level 2, Dr. Brugge and Dr. Zoeller aimed to evaluate whether BCL-2/XL inhibition by ABT-263 (navitoclax) could enhance the effectiveness of T-DM1 in HER2-expressing breast cancers.
Using five HER2-expressing patient-derived xenograft (PDX) mouse models developed from primary and metastatic breast tumors with and without prior treatment exposure, a combination of T-DM1 and ABT-263 was compared to treatment with either agent alone. Results from this study were recently published in Molecular Cancer Therapeutics and demonstrated that two of the treatment-exposed metastatic PDX-models, PDX12 and PDX8, had the most robust responses to combination therapy. Treatment with combination therapy in PDX12 and PDX8 mouse models significantly enhanced tumor killing and resulted in a reduction in tumor size compared to either agent alone. Microscopic analysis of PDX12 and PDX8 tumor sections revealed that combination treatment resulted in a dramatic reduction in the number of invasive tumor cells present, whereas single agent treatment alone had little to no effect. Tumors from PDX model BCM-3963 (treatment-naïve primary breast tumor) were responsive to T-DM1 alone, but little to no additive effect on tumor size was observed following combination treatment. However, upon microscopic analysis of BCM-3963 tumor sections, an increase in cancer cell killing was seen when T-DM1 was administered in combination with ABT-263, suggesting that ABT-263 enhanced the cytotoxicity of T-DM1.
Although BCL-2 inhibitors have shown promise in clinical trials, their use has been limited due to thrombocytopenia (platelet reduction) observed in patients. To overcome this treatment-associated side effect, the team devised a pulsed dosing regimen where ABT-263 was administered in intervals rather than continuously. T-DM1 treatment given with pulsed ABT-263 did not reduce the pre-clinical effectiveness of combination treatment in PDX12 and PDX8 tumors, and importantly, mitigated the platelet side effects associated with ABT-263.
These results demonstrate for the first time that the effectiveness of T-DM1 can be enhanced by inhibition of BCL-2/XL using an orally available and clinically tested compound (navitoclax), and provide evidence for a translational paradigm involving ADCs together with anti-apoptotic inhibitors in order to ameliorate the systemic toxicities associated with these therapies.
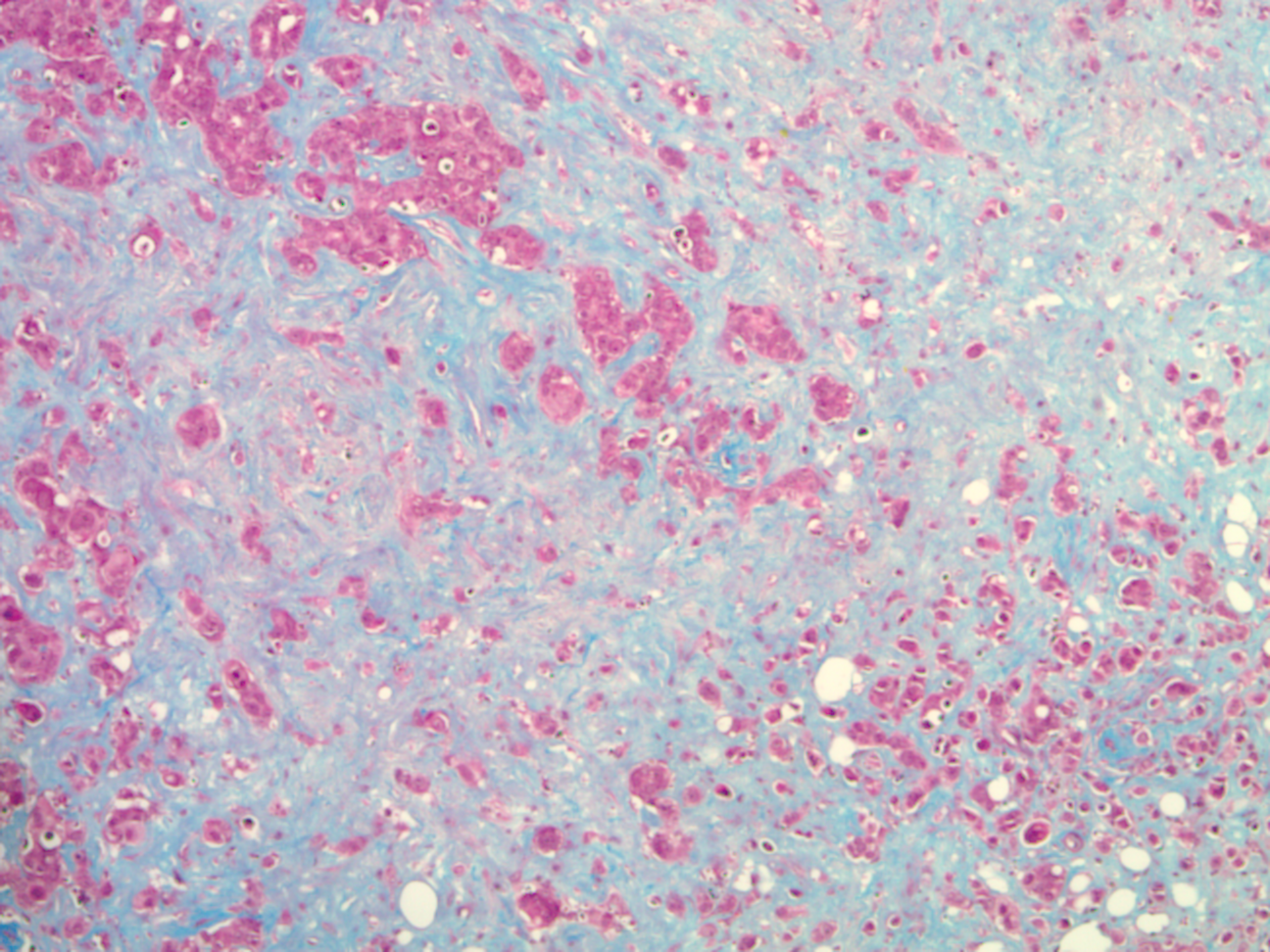
Pre-clinical in vivo treatment response associated with navitoclax+T-DM1 were assessed by Masson's trichrome stain and were characterized by the significant elimination of tumor cells (red) and the emergence of stromal content (blue)
Publication:
Zoeller JJ, Vagodny A, Taneja K, et al. 2019. Neutralization of BCL-2/XL enhances the cytotoxicity of T-DM1 in vivo. Mol Cancer Ther. 18(6):1115-1126.
Link:
Last updated Thursday, February 19, 2026
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
