


- Development of the BreastCancerTrials.org Website
- Characterization of Human Mammary Epithelial Stem Cells
- The Impact of Gestational and Postnatal Dietary Exposures on Metabolic Programming and Breast Cancer Risk
- The Influence of Neuronal Activity on Breast Cancer Metastasis
- Integrated Molecular Imaging and Therapy for Breast Cancer
- Fibrinogen-Mediated Mechanisms of Breast Cancer Metastasis
- Using MRI Technology to Improve Treatment Strategies for Breast Cancer
- Early Life Processes, Endocrine Mediators, and Number of Susceptible Cells in Relation to Breast Cancer Risk
- Converting Bcl-2 from a Protector to a Killer
Development of the BreastCancerTrials.org Website
Posted December 14, 2009
Laura Esserman, M.D., University of California, San Francisco
 The Fiscal Year 1995 (FY95) Breast Cancer Research Program Cancer Center Award to Dr. Laura Esserman from the University of California, San Francisco (UCSF), provided the first funds to set up an initial clinical trial database for UCSF. The website, bcc-ct.his.ucsf.edu, was initiated as the Breast Cancer Care Center Clinical Trials Website and access to it was limited to UCSF physicians and patients. In 1998, two breast cancer survivors, Joan Schreiner and Joanne Tyler, envisioned a clinical trials matching service that would be available, at no cost, to all patients, would have a patient-centered design, incorporate a non-profit status, would be trusted/non-biased, and would be affiliated with an academic medical center. After meeting with UCSF breast cancer specialists, including Dr. Esserman, this team established a goal of increasing participation in breast cancer clinical trials while helping patients make informed decisions about trial participation. A FY02 Breast Cancer Center of Excellence Award to Dr. Esserman provided project management for development of BreastCancerTrials.org, a new, patient-centered web service designed to help breast cancer patients find appropriate clinical trials by matching their self-reported breast cancer histories to the eligibility criteria of listed trials. The website, https://www.breastcancertrials.org/, was initially launched in a regional Bay Area pilot in June 2005 and was so successful that it opened to the public in 2008 as a nationwide, nonprofit service.
The Fiscal Year 1995 (FY95) Breast Cancer Research Program Cancer Center Award to Dr. Laura Esserman from the University of California, San Francisco (UCSF), provided the first funds to set up an initial clinical trial database for UCSF. The website, bcc-ct.his.ucsf.edu, was initiated as the Breast Cancer Care Center Clinical Trials Website and access to it was limited to UCSF physicians and patients. In 1998, two breast cancer survivors, Joan Schreiner and Joanne Tyler, envisioned a clinical trials matching service that would be available, at no cost, to all patients, would have a patient-centered design, incorporate a non-profit status, would be trusted/non-biased, and would be affiliated with an academic medical center. After meeting with UCSF breast cancer specialists, including Dr. Esserman, this team established a goal of increasing participation in breast cancer clinical trials while helping patients make informed decisions about trial participation. A FY02 Breast Cancer Center of Excellence Award to Dr. Esserman provided project management for development of BreastCancerTrials.org, a new, patient-centered web service designed to help breast cancer patients find appropriate clinical trials by matching their self-reported breast cancer histories to the eligibility criteria of listed trials. The website, https://www.breastcancertrials.org/, was initially launched in a regional Bay Area pilot in June 2005 and was so successful that it opened to the public in 2008 as a nationwide, nonprofit service.
BreastCancerTrials.org
Links:
Public and Technical Abstracts: Blueprint for Regional Excellence in Breast Cancer Care
Characterization of Human Mammary Epithelial Stem Cells
Posted November 19, 2009
Peter Eirew, British Columbia Cancer Agency, Vancouver, British Columbia
 Normal human tissues have been found to contain very small numbers of stem cells, each of which has the ability to regenerate all of the different cell types and functional elements of the original tissue. There is a growing body of evidence supporting a central role for stem cells in the development of breast cancer, underscoring the importance of better understanding the biology of these cells in the human breast. Peter Eirew from the British Columbia Cancer Agency received a Department of Defense Fiscal Year 2006 Breast Cancer Research Program Predoctoral Traineeship Award to develop a method for isolating and characterizing the rare populations of stem cells found in normal and malignant human mammary tissues. In his PhD thesis project mentored by Dr. Connie Eaves, Mr. Eirew developed and validated a robust, reproducible, and specific method to isolate and characterize normal human mammary stem cells using a xenotransplant model. Suspensions of dissociated normal human mammary cells were transplanted into immunodeficient mice and allowed to grow for several weeks. A rare subset of cells was found to possess in vivo regenerative properties, forming structures that closely resemble normal human mammary glands with an interior surface of polarized luminal cells that could be induced to produce milk and a secondary layer of myoepithelial cells, surrounded by an outer basement membrane. Regenerated mammary glands also contained mammary progenitor cells capable of generating colonies in vitro, as well as re-transplantable stem cells, indicating a regenerative process that reproduces a tissue hierarchy. The number of daughter progenitor cells detected after 4 weeks in vivo serves as a sensitive and objective retrospective measure of the stem cell content of the cells transplanted, thus providing a method to quantify the stem cell frequency in any population by limiting dilution analysis. By further analyzing the regenerative properties of phenotypically distinct mammary subpopulations, Mr. Eirew identified a preliminary set of human mammary stem cell markers that can be used to sort and purify these rare cells directly from normal mammary tissue. This project lays the ground for future work to characterize the biological differences between normal human mammary stem cells and breast cancer stem cells, with the goal of elucidating how they might regulate breast cancer.
Normal human tissues have been found to contain very small numbers of stem cells, each of which has the ability to regenerate all of the different cell types and functional elements of the original tissue. There is a growing body of evidence supporting a central role for stem cells in the development of breast cancer, underscoring the importance of better understanding the biology of these cells in the human breast. Peter Eirew from the British Columbia Cancer Agency received a Department of Defense Fiscal Year 2006 Breast Cancer Research Program Predoctoral Traineeship Award to develop a method for isolating and characterizing the rare populations of stem cells found in normal and malignant human mammary tissues. In his PhD thesis project mentored by Dr. Connie Eaves, Mr. Eirew developed and validated a robust, reproducible, and specific method to isolate and characterize normal human mammary stem cells using a xenotransplant model. Suspensions of dissociated normal human mammary cells were transplanted into immunodeficient mice and allowed to grow for several weeks. A rare subset of cells was found to possess in vivo regenerative properties, forming structures that closely resemble normal human mammary glands with an interior surface of polarized luminal cells that could be induced to produce milk and a secondary layer of myoepithelial cells, surrounded by an outer basement membrane. Regenerated mammary glands also contained mammary progenitor cells capable of generating colonies in vitro, as well as re-transplantable stem cells, indicating a regenerative process that reproduces a tissue hierarchy. The number of daughter progenitor cells detected after 4 weeks in vivo serves as a sensitive and objective retrospective measure of the stem cell content of the cells transplanted, thus providing a method to quantify the stem cell frequency in any population by limiting dilution analysis. By further analyzing the regenerative properties of phenotypically distinct mammary subpopulations, Mr. Eirew identified a preliminary set of human mammary stem cell markers that can be used to sort and purify these rare cells directly from normal mammary tissue. This project lays the ground for future work to characterize the biological differences between normal human mammary stem cells and breast cancer stem cells, with the goal of elucidating how they might regulate breast cancer.
Publications:
Eirew P, Stingl J, Raouf A, et al. 2008. A method for quantifying normal human mammary epithelial stem cells with in vivo regenerative ability. Nature Medicine 14:1384-1389.
Links:
Public and Technical Abstracts: Characterization of Human Mammary Epithelial Stem Cells
The Impact of Gestational and Postnatal Dietary Exposures on Metabolic Programming and Breast Cancer Risk
Posted October 23, 2009
Robin Fuchs-Young, Ph.D., University of Texas M. D. Anderson Cancer Center, Houston, Texas
 Several lines of scientific evidence suggest that dietary exposures during windows of critical fetal development can permanently affect energy balance and metabolism of offspring. Children of obese mothers are more likely to have an abnormal birth weight, which is associated with an increased risk of obesity and breast cancer later in life. In studies using animal models, the offspring of mice who were fed a high carbohydrate diet suffered from sustained hyperinsulinemia through adulthood. Moreover, these metabolic changes can be further passed onto future progeny, suggesting that perinatal environmental exposures can induce adaptive changes at the genetic level and at a very early age.
Several lines of scientific evidence suggest that dietary exposures during windows of critical fetal development can permanently affect energy balance and metabolism of offspring. Children of obese mothers are more likely to have an abnormal birth weight, which is associated with an increased risk of obesity and breast cancer later in life. In studies using animal models, the offspring of mice who were fed a high carbohydrate diet suffered from sustained hyperinsulinemia through adulthood. Moreover, these metabolic changes can be further passed onto future progeny, suggesting that perinatal environmental exposures can induce adaptive changes at the genetic level and at a very early age.
Dr. Robin Fuchs-Young at the University of Texas M. D. Anderson Cancer Center received a Fiscal Year 2007 Breast Cancer Research Program Idea Award to investigate the impact of gestational and postnatal dietary exposures on metabolic programming and breast cancer risk. She hypothesizes that metabolic programming, stemming from gestational and/or perinatal exposure to maternal hyperglycemia and hyperinsulinemia, influences the offspring's susceptibility to mammary carcinogenesis. Further, the researcher suspects that mutations in mitochondrial genes, induced by elevated glucose metabolism in mammary cells, are a potential mechanism underlying this increased susceptibility to breast cancer.
Dr. Fuchs-Young is evaluating the effects of maternal obesity on mammary development and susceptibility to mammary cancer of the exposed offspring. Tissues and tumors, excised from experimental mice will be examined at various stages of development for oxidative mitochondrial DNA damage and mutations. The ability of high glucose exposure to impact mitochondrial function and tumorigenicity will also be assessed in immortalized, but untransformed, breast epithelial cells in vitro and in vivo. This study will challenge the long-standing paradigm that obesity is only a risk factor for breast cancer in post-menopausal women.
The Influence of Neuronal Activity on Breast Cancer Metastasis
Posted October 9, 2009
Anna Majewska, Ph.D., and Edward Brown, Ph.D., University of Rochester Medical Center
 Few effective therapies exist to treat breast cancer brain metastases and, as a result, they are a primary cause of mortality in breast cancer patients. Dr. Anna Majewska and Dr. Edward Brown, from the University of Rochester Medical Center in New York, received an FY06 Synergistic Idea Award to investigate a novel hypothesis that, if successful, could greatly improve our ability to both predict and prevent tumor metastasis in the brain. Specifically, they expect that normal neuronal mechanisms that alter synaptic motility and the extracellular matrix (ECM) may also affect the ability of breast tumor cells to form metastases in the brain. Nude mice are being treated with the clinically approved neuronal stimulants caffeine, methylphenidate, and modafinil to examine the effect of these drugs on brain metastasis. Interestingly, preliminary data has shown that caffeine treatment reduced the brain tumor burden twofold when compared to controls. Further, caffeine-treated mice had a much larger proportion of white matter tumors than controls, supporting the idea that caffeine can cause differences in the location of tumor seeding. To understand the underlying mechanisms involved, the scientists are also examining the involvement of the serine proteinase tissue plasminogen activator (tPA) and changes in the brain ECM in the metastasis-hindering effects of these drugs. This work could establish a new paradigm in metastatic control whereby interventions that are able to cross the blood-brain barrier (unlike standard chemotherapeutics) and modify neuronal activity in normal brain tissue (rather than attacking tumor cells directly) can be used to inhibit tumor growth and invasion.
Few effective therapies exist to treat breast cancer brain metastases and, as a result, they are a primary cause of mortality in breast cancer patients. Dr. Anna Majewska and Dr. Edward Brown, from the University of Rochester Medical Center in New York, received an FY06 Synergistic Idea Award to investigate a novel hypothesis that, if successful, could greatly improve our ability to both predict and prevent tumor metastasis in the brain. Specifically, they expect that normal neuronal mechanisms that alter synaptic motility and the extracellular matrix (ECM) may also affect the ability of breast tumor cells to form metastases in the brain. Nude mice are being treated with the clinically approved neuronal stimulants caffeine, methylphenidate, and modafinil to examine the effect of these drugs on brain metastasis. Interestingly, preliminary data has shown that caffeine treatment reduced the brain tumor burden twofold when compared to controls. Further, caffeine-treated mice had a much larger proportion of white matter tumors than controls, supporting the idea that caffeine can cause differences in the location of tumor seeding. To understand the underlying mechanisms involved, the scientists are also examining the involvement of the serine proteinase tissue plasminogen activator (tPA) and changes in the brain ECM in the metastasis-hindering effects of these drugs. This work could establish a new paradigm in metastatic control whereby interventions that are able to cross the blood-brain barrier (unlike standard chemotherapeutics) and modify neuronal activity in normal brain tissue (rather than attacking tumor cells directly) can be used to inhibit tumor growth and invasion.
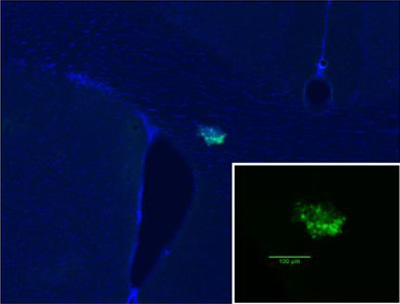
A breast tumor metastasis within the rodent brain. Green represents the fluorescent breast cancer cells, while blue is a cellular DAPI stain that reveals brain morphology. The inset shows the tumor at a higher magnification.
Integrated Molecular Imaging and Therapy for Breast Cancer
Posted July 2, 2009
Balaji Panchapakesan, Ph.D., Delaware Biotechnology Institute,
University of Delaware, Newark, Delaware
 Many researchers are investigating novel and innovative ways to improve breast cancer detection. The field of nanotechnology offers a new dimension of possibilities in this realm of research. Dr. Balaji Panchapakesan, of the University of Delaware, is a recipient of Department of Defense Fiscal Year 2002 (FY02) and FY05 Breast Cancer Research Program (BCRP) Concept Awards that focused on developing carbon nanotubes as the basis for integrating molecular imaging and therapy in one single step.
Many researchers are investigating novel and innovative ways to improve breast cancer detection. The field of nanotechnology offers a new dimension of possibilities in this realm of research. Dr. Balaji Panchapakesan, of the University of Delaware, is a recipient of Department of Defense Fiscal Year 2002 (FY02) and FY05 Breast Cancer Research Program (BCRP) Concept Awards that focused on developing carbon nanotubes as the basis for integrating molecular imaging and therapy in one single step.
Dr. Panchapakesan created a nanotube with a heat-sensitive core that can be activated by heat pulses. If heated nanotubes are attached to or taken up by cells, they will kill the cells. Breast cancer cells often overexpress Her2 and IGF1R receptors. For that reason, Dr. Panchapakesan attached antibodies specific to Her2 and IGF1R receptors to the nanotubes, thereby allowing the nanotubes to specifically target breast cancer cells. The final step in creating a "three-in-one" molecule was to conjugate gold nanoparticles with the nanotube-bound antibodies. Gold nanoparticles change color from red to blue when they aggregate or interact with adjacent nanoparticles. The amalgamation of nanotube-antibody-gold particles would allow this molecule to target specific surface receptors such as Her2 in breast cancer cells, image the cancer cells when color change results from aggregation of the gold nanoparticles, and kill the cancerous cells with heat pulses that cause the core of the nanotube to burst. This innovative technology may eventually lead to the first clinical application of a single molecule that completes targeting, imaging, and therapy all in one step. Dr. Panchapakesan writes, "These tools can also be applied to traditional surgical oncology to improve the probability of killing cancer cells whose microscopic foci are often left behind in surgery."
Publications:
Shao N, Lu S, Wickstorm E, et al. 2008. Integrated molecular targeting of IGF1R and HER2 surface receptors and destruction of breast cancer cells using single wall carbon nanotubes. Nanotechnology 18(31):315101.
Links:
Fibrinogen-Mediated Mechanisms of Breast Cancer Metastasis
Posted June 1, 2009
Patricia J. Simpson-Haidaris, Ph.D., University of Rochester, New York
 Systemic inflammation is clearly linked to an adverse prognosis in various cancers, including breast cancer, and is characterized by elevated expression of pro-inflammatory mediators and plasma proteins including fibrinogen. Fibrinogen is a key component of the tumor stroma-extracellular matrix, and it promotes tumor cell metastasis following inflammation; however, the molecular mechanisms by which this fibrinogen-mediated metastasis occurs are not fully understood. Dr. Patricia Simpson-Haidaris from the University of Rochester received a Department of Defense Fiscal Year 2005 Breast Cancer Research Program Concept Award to investigate the role of fibrinogen in breast cancer metastasis.
Systemic inflammation is clearly linked to an adverse prognosis in various cancers, including breast cancer, and is characterized by elevated expression of pro-inflammatory mediators and plasma proteins including fibrinogen. Fibrinogen is a key component of the tumor stroma-extracellular matrix, and it promotes tumor cell metastasis following inflammation; however, the molecular mechanisms by which this fibrinogen-mediated metastasis occurs are not fully understood. Dr. Patricia Simpson-Haidaris from the University of Rochester received a Department of Defense Fiscal Year 2005 Breast Cancer Research Program Concept Award to investigate the role of fibrinogen in breast cancer metastasis.
To determine the effects of fibrinogen on vascular permeability, monolayers of human umbilical vein endothelial cells (HUVEC) and HMEC-1 microvascular endothelial cells were exposed to increasing concentrations of fibrinogen, and the resulting flux of FITC-Dextran across the endothelial cell (EC) barrier was measured. Fibrinogen induced the permeability of both HUVEC and HMEC-1 monolayers, with maximum permeability observed in the presence of 150-300 nM fibrinogen. Dr. Simpson-Haidaris then investigated the effects of a specific fibrinogen domain, Fg-β15-42, which binds to the cell-cell adhesion molecule VE-cadherin on endothelial cells and is known to promote cell adhesion, proliferation, and migration during angiogenesis and tumor growth. Antibodies against Fg-β15-42 or the VE-cadherin extracellular domain inhibited the effects of fibrinogen on permeability, resulting in reduced FITC-Dextran flux across the endothelium. Fibrinogen treatment was also found to redistribute VE-cadherin from the cell surface into the cytoplasm. Transcellular migration assays demonstrated that fibrinogen and Fg-β15-42 each induced the transendothelial migration of MDA-MB-231 and MCF-7 breast cancer cells, but not non-malignant MCF-10A breast epithelial cells. Antibodies against the Fg-β15-42 domain or VE-cadherin blocked the fibrinogen-induced transendothelial migration of breast cancer cells. These results show that Fg-β15-42 may be an exciting new molecular target for therapeutic development against breast cancer metastasis.

Publication of the International Union Against Cancer
Published for the UICC by Wiley-Liss, Inc
© 2009 UICC
Fibrinogen-endothelial cell interactions. Endothelial cells grown on gelatin-coated glass coverslips were treated with purified plasma fibrinogen conjugated to a green fluorescent dye. The cells were fixed, permeabilized and stained with antibodies to fibroblast growth factor-2 (red fluorescence depicting the endothelial cells). The coverslip was mounted "upside down" on a microscope slide such that the basolateral aspect and the subendothelial extracellular matrix appear on "top" of the cells. Epifluorescence microscopy revealed that plasma fibrinogen binds to endothelial cell-cell junctions in a punctate pattern (green fluorescence), consistent with fibrinogen binding to its intercellular surface receptor, vascular endothelial cadherin (VE-cadherin). Fibrinogen also assembles to a limited extent into the subendothelial fibrillar extracellular matrix as denoted by the strands of green fluorescence. The accompanying manuscript in the International Journal of Cancer shows that fibrinogen induction of endothelial barrier permeability and enhanced transendothelial migration of malignant breast cancer cells requires fibrinogen-VE-cadherin binding interactions.
Publication:
Sahni A, Arévalo MT, Sahni SK, and Simpson-Haidaris PJ. 2009. The VE-cadherin binding domain of fibrinogen induces endothelial barrier permeability and enhances transendothelial migration of malignant breast epithelial cells. Int J Cancer (in press; cover photo).
Links:
Public and Technical Abstracts: Fibrin(ogen)-Mediated Pathways of Breast Cancer Metastasis
Using MRI Technology to Improve Treatment Strategies for Breast Cancer
Posted March 31, 2009
Dawen Zhao, M.D., Ph.D., University of Texas Southwestern Medical Center at Dallas, Texas
 Dr. Dawen Zhao, of the University of Texas Southwestern Medical Center at Dallas, is a Department of Defense Breast Cancer Research Program Idea Awardee for fiscal year 2002 (FY02) and FY07. Dr. Zhao specializes in the study of tumor physiology using novel magnetic resonance imaging (MRI) techniques. In his FY02 Idea Award project, Dr. Zhao used 19F (FREDOM) (Fluorocarbon Relaxometry using Echo planar imaging for Dynamic Oxygen Mapping) MRI to monitor tumor oxygenation following treatment with combretastatin A-4-phosphate (CA4P), a vascular targeting agent. Drugs like CA4P that effectively target the tumor vasculature are promising anti-cancer therapies because they can disrupt the flow of nutrients to thousands of tumor cells, thus killing the cells and inhibiting the growth of new blood vessels (angiogenesis). However, because small rings of viable tumor tissue will remain, radiation treatment may also be needed. The effectiveness of radiation treatment depends upon the oxygen levels of the tumor tissue. Therefore, Dr. Zhao has used 19F MRI to monitor the dynamics of tumor oxygen levels for the development of an optimal strategy that combines both radiation and CA4P therapy for the treatment of breast cancer. Seven unique treatment strategies were tested in 13762NF rat breast cancer tumors. The most significant delay in tumor development was seen in rats treated with CA4P (30/mg/kg) plus a 24-hour delay before treatment with radiation (5Gy) and oxygen (100% +1% isofluorane). MRI findings were found to correlate with histological studies of tumor hypoxia and tumor vessel perfusion, which supports the use and accuracy of this technology to study tumor pathophysiology.
Dr. Dawen Zhao, of the University of Texas Southwestern Medical Center at Dallas, is a Department of Defense Breast Cancer Research Program Idea Awardee for fiscal year 2002 (FY02) and FY07. Dr. Zhao specializes in the study of tumor physiology using novel magnetic resonance imaging (MRI) techniques. In his FY02 Idea Award project, Dr. Zhao used 19F (FREDOM) (Fluorocarbon Relaxometry using Echo planar imaging for Dynamic Oxygen Mapping) MRI to monitor tumor oxygenation following treatment with combretastatin A-4-phosphate (CA4P), a vascular targeting agent. Drugs like CA4P that effectively target the tumor vasculature are promising anti-cancer therapies because they can disrupt the flow of nutrients to thousands of tumor cells, thus killing the cells and inhibiting the growth of new blood vessels (angiogenesis). However, because small rings of viable tumor tissue will remain, radiation treatment may also be needed. The effectiveness of radiation treatment depends upon the oxygen levels of the tumor tissue. Therefore, Dr. Zhao has used 19F MRI to monitor the dynamics of tumor oxygen levels for the development of an optimal strategy that combines both radiation and CA4P therapy for the treatment of breast cancer. Seven unique treatment strategies were tested in 13762NF rat breast cancer tumors. The most significant delay in tumor development was seen in rats treated with CA4P (30/mg/kg) plus a 24-hour delay before treatment with radiation (5Gy) and oxygen (100% +1% isofluorane). MRI findings were found to correlate with histological studies of tumor hypoxia and tumor vessel perfusion, which supports the use and accuracy of this technology to study tumor pathophysiology.
Brain metastasis has emerged in recent years as a common cause of death in breast cancer patients. Dr. Zhao's FY07 Idea Award was dedicated to the study of intracranial hypoxia using imaging approaches that his laboratory recently developed based on blood oxygenation level-dependent (BOLD) MRI and dynamic contrast enhanced (DCE) MRI. Tumor hypoxia is well known to indicate poor prognosis and is associated with an aggressive clinical and biological phenotype. Dr. Zhao will use these MRI technologies to understand how intracranial tumor hypoxia correlates with blood brain barrier permeability and tumor aggressiveness, as a means of improving outcomes for breast cancer patients suffering from brain metastasis.
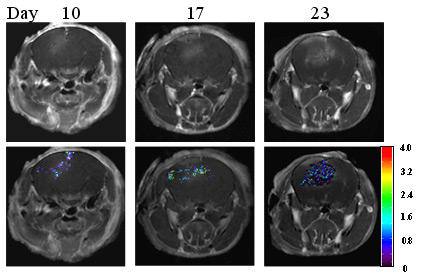
Dynamic MRI evaluation of blood brain barrier (BBB) function in a mouse model of breast tumor brain metastasis. Sequential MRI scans were performed at different time points after intracranial tumor implantation. T1-weighted contrast enhanced MRI showed a tumor growing intracranially (Top row). The BBB permeability is evaluated by calculating the constant of contrast exchange rate Kep based on dynamic contrast enhanced (DCE) MRI. The Kep maps overlapping on the anatomic images revealed development of BBB disruption and heterogeneity of BBB permeability within the tumor (Bottom row). This information will be useful for adjuvant chemotherapy or targeted molecular therapy.
Publications:
Zhao D, Richer E, Antich PP, et al. 2008. Antivascular effects of combretastatin A4 phosphate in breast cancer xenograft assessed using dynamic bioluminescence imaging and confirmed by MRI. FASEB Journal 22:2445-2451.
Zhao D, Jiang L, Hahn EW, et al. 2005. Tumor physiological response to combretastatin A4 phosphate assessed by MRI. International Journal of Radiation Oncology, Biology, Physics 62:872-880.
Links:
Early Life Processes, Endocrine Mediators, and Number of Susceptible Cells in Relation to Breast Cancer Risk
Posted February 17, 2009
Dimitrios Trichopoulos, M.D., Ph.D., Harvard College, Boston, Massachusetts
 Dimitrios Trichopoulos, a fiscal year 2004 Breast Cancer Research Program (BCRP) Innovator Award recipient, was a 2007 recipient of a Medal of Honor from the International Agency for Research on Cancer (IARC), which is part of the World Health Organization. He was honored for his research in cancer etiology, including work funded by his BCRP Innovator Award in which he examines the role of perinatal events and conditions in the etiology of breast cancer in adult life. Dr. Trichopoulos' research project is based on the hypothesis that breast cancer risk is a function of the number of mammary gland cells at risk of transformation. He postulates that this number is likely to be reflected in birth size and rate of postnatal growth and, in turn, correlates to mammary gland mass and breast cancer risk in adult life. Dr. Trichopoulos is currently undertaking five complementary studies, three of which focus on perinatal characteristics in relation to mammary gland mass and breast cancer risk, and two which examine the relationship of pregnancy hormones with birth characteristics, as well as cellular populations likely to have mammary stem cell potential. Analysis of previous data on hematopoietic stem cells in cord blood showed that, among term newborns with a normal-to-high birth weight, birth weight was significantly positively associated with stem cell measurements, supporting a role for the stem cell pool in the risk of cancer. Identifying early life risk factors that contribute to the development of breast cancer could aid in better medical surveillance of females at risk, and provide earlier and possibly better opportunities for prevention of the disease.
Dimitrios Trichopoulos, a fiscal year 2004 Breast Cancer Research Program (BCRP) Innovator Award recipient, was a 2007 recipient of a Medal of Honor from the International Agency for Research on Cancer (IARC), which is part of the World Health Organization. He was honored for his research in cancer etiology, including work funded by his BCRP Innovator Award in which he examines the role of perinatal events and conditions in the etiology of breast cancer in adult life. Dr. Trichopoulos' research project is based on the hypothesis that breast cancer risk is a function of the number of mammary gland cells at risk of transformation. He postulates that this number is likely to be reflected in birth size and rate of postnatal growth and, in turn, correlates to mammary gland mass and breast cancer risk in adult life. Dr. Trichopoulos is currently undertaking five complementary studies, three of which focus on perinatal characteristics in relation to mammary gland mass and breast cancer risk, and two which examine the relationship of pregnancy hormones with birth characteristics, as well as cellular populations likely to have mammary stem cell potential. Analysis of previous data on hematopoietic stem cells in cord blood showed that, among term newborns with a normal-to-high birth weight, birth weight was significantly positively associated with stem cell measurements, supporting a role for the stem cell pool in the risk of cancer. Identifying early life risk factors that contribute to the development of breast cancer could aid in better medical surveillance of females at risk, and provide earlier and possibly better opportunities for prevention of the disease.
Selected Publications:
Trichopoulos D, Adami H, Ekbom A, Hsieh C-C, Lagiou P. 2008. Early life events and conditions and breast cancer risk: from epidemiology to etiology. Int. J. Cancer 122:481-485
Lagiou P, HsiehC-C, Trichopoulos D, Adami H-O, Hal P, Chie L and Ekbom A. 2008. Neonatal growth and breast cancer risk in adulthood. British J. Cancer 99:1544-1548.
Converting Bcl-2 from a Protector to a Killer
Posted January 23, 2009
Arnold C. Satterthwait, Ph.D., Burnham Institute for Medical Research, La Jolla, California
Bcl-2 is a member of the large Bcl-2 family of proteins that regulate programmed cell death or apoptosis. While Bcl-2 typically functions as a protective, anti-apoptotic protein, it is converted to a pro-apoptotic killer protein by a member of the nuclear receptor superfamily, Nur77, (also called TR3 or RGFI-B) that binds its' loop. Dr. Arnold Satterthwait, who received a Fiscal Year 2002 Idea Award, and his colleagues at the Burnham Institute sought to develop short Nur77 peptides that could potentially convert Bcl-2 to its pro-apoptotic form thus reversing the resistance of breast cancer cells to chemotherapy.
Peptides corresponding to a Nur77 fragment that interacts with Bcl-2 were synthesized, linked to a cell-permeable polyarginine peptide and tested for pro-apoptotic effects on cancer cells with high Bcl-2 levels. A 9-amino acid Nur77-derived Bcl-2 converting peptide (NuBCP-9) was identified that showed potent pro-apoptotic effects on breast cancer cells in culture and in animals, but not on normal primary mammary epithelial cells. The NuBCP-9 enantiomer, created by replacing L-amino acids with D-amino acids, was equally functional at inducing apoptosis. Further experiments confirmed that NuBCP-9s induced apoptosis through a Bcl-2-dependent mechanism by binding its loop, which displaced a connecting N-terminal helix. This conformational change both neutralized Bcl-2's inhibition of Bax-mediated apoptosis, and exposed a latently, pro-apoptotic BH3 domain that acted by an indirect mechanism to inhibit the anti-apoptotic Bcl-xL. The majority of human-cancer-associated proteins like Bcl-2 are predicted to have large, structurally adaptive loops which can be functionally advantageous and account for enantiomer activity. D-peptides, unlike normal peptides, are resistant to proteolysis which can be advantageous for drug design. Thus the observation that a peptide enantiomer can act by binding unstructured loops which are characteristic of cancer-associated proteins may have wider significance. Taken together, these results not only provide new mechanistic insight into Bcl-2 conversion, but also identify NuBCP-9 and its enantiomer as new drug leads that have significant potential as breast cancer therapeutics.
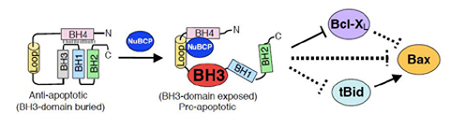
Flipping the Bcl-2 switch. NuBCP-9 and its enantiomer bind a large, natively disordered loop on Bcl-2 that displaces the BH4 domain, exposing Bcl-2's latently pro-apoptotic BH3 domain. This leads to both the release of pro-apoptotic BH3-only activators (e.g., tBid) from Bcl-2 and the inhibition of other anti-apoptotic Bcl-2 family proteins (e.g., Bcl-xL) by the exposed BH3 domain. Displaced pro-apoptotic multi-BH3 family members (e.g., Bax) can be oligomerized on mitochondria by activators to release apoptogenic factors that induce apoptosis.
Publication:
Kolluri SK, Zhu X, Zhou X, Lin B, Chen Y, Sun K, Tian X, Town J, Cao X, Lin F, Zhai D, Kitada S, Luciano F, O'Donnell E, Cao Y, He F, Lin J, Reed JC, Satterthwait AC, Zhang XK. 2008. A short Nur77-derived peptide converts Bcl-2 from a protector to a killer. Cancer Cell 14:1-14.
Highlighted:
Nature Biotechnology. 2008. 26: 1250
Nature Reviews Drug Discovery. 2008. 7: 977
News and Views:
Martz, L. 2008. Bcl-2 double take. SciBX 1(39): 1-4
Qi, B., Hardwick, J.M. 2008. Bcl-2 turns deadly. Nature Chemical Biology 4: 722-723














