Breast Cancer
Neutrophil Extracellular Traps Produced During Inflammation Awaken Dormant Cancer Cells



Posted April 23, 2019
Mikala Egeblad, Ph.D., Cold Spring Harbor Laboratory
For virtually all breast cancer patients who succumb to their disease, death results from metastases that are generally resistant to therapies, even after successful treatment of the primary tumor. Although the triggers that reawaken dormant tumor cells are poorly understood, research has found that chronic inflammation adjacent to dormant disseminated tumor cells (DTCs), and changes in the local extracellular matrix (ECM) surrounding the tumor can promote the formation of lethal metastases. Neutrophils, key inflammatory response cells, have been identified as fundamental to the reawakening of dormant DTCs, although the processes underlying this remains mysterious. Uncovering the precise inflammatory mechanisms utilized by neutrophils that lead to the reawakening of DTCs could reveal new therapeutic targets for the prevention and treatment of lethal metastatic disease. With a Fiscal Year 2013 Breast Cancer Research Program Era of Hope Scholar Award, Dr. Egeblad elegantly uncovered how neutrophil-mediated chronic inflammation encourages the reawakening of breast cancer cells to form lung metastases and then demonstrated these processes could be blocked.
In a recent manuscript published in Science, Dr. Egeblad’s team established a mouse model of chronic inflammation where dormant breast cancer cells awaken, multiply, and form metastases in the lungs. Her research revealed that the process relies on the activity of nearby neutrophils and is dependent on the formation of neutrophil extracellular traps (NETs). NETs are mixtures of DNA and enzymes released by neutrophils, which can modify the extracellular environment. Using in vitro models, isolated NETs were found to be capable of promoting growth in dormant breast cancer cells.

Lab team photo: Dr. Egeblad (back row, second on the right)
Cells sense changes in the ECM largely through integrins, protein complexes found on the surface of cells. Cancer cell reawakening is known to be regulated by these proteins, and the research group identified a specific integrin complex (α3β1) that is necessary for the NET-induced reawakening. Further, the team determined that NETs cause a unique modification to the core ECM protein, laminin, which is recognized by the cancer cells to stimulate growth. In search of a way to exploit this new knowledge to clinically prevent recurrence, Dr. Egeblad’s team generated an antibody specific for NET enzyme modified laminin, Ab28. Using Ab28 in in vitro models, they demonstrated that NET-induced reawakening of breast cancer cells could be blocked. In the mouse, Ab28 bound laminin only after it had been modified by chronic inflammation, and was found in close proximity to NETs and proliferating cancer cells in the lung as determined by fluorescent microscopy. These findings suggest that Ab28 would likely not interfere with the biology of normal laminin expressed throughout the body. Most importantly, a version of Ab28 with low immunogenic potential blocked inflammation-induced awakening of dormant DTCs in the lung without proving to be lethal.
Results from this study demonstrate that dormant DTC reawakening can be therapeutically blocked thus potentially preventing the development of lethal metastatic disease. This work exemplifies Dr. Egeblad’s goal to target the microenvironment as a means of combating breast cancer recurrence, and she has developed a tool that may crack the code for patients with chronic lung inflammation who develop recurrent disease as a result. Her study demonstrated that inflammation directly affects the microenvironment in the lung, changing the extracellular matrix to provide a growth signal to local dormant breast cancer cells. Most impressively, this information was harnessed to provide a proof-of-concept antibody therapeutic for inhibiting this phenomenon. These findings provide an exciting new path forward for breast cancer patients, particularly those suffering from chronic lung inflammation, to prevent the onset of recurrent disease.
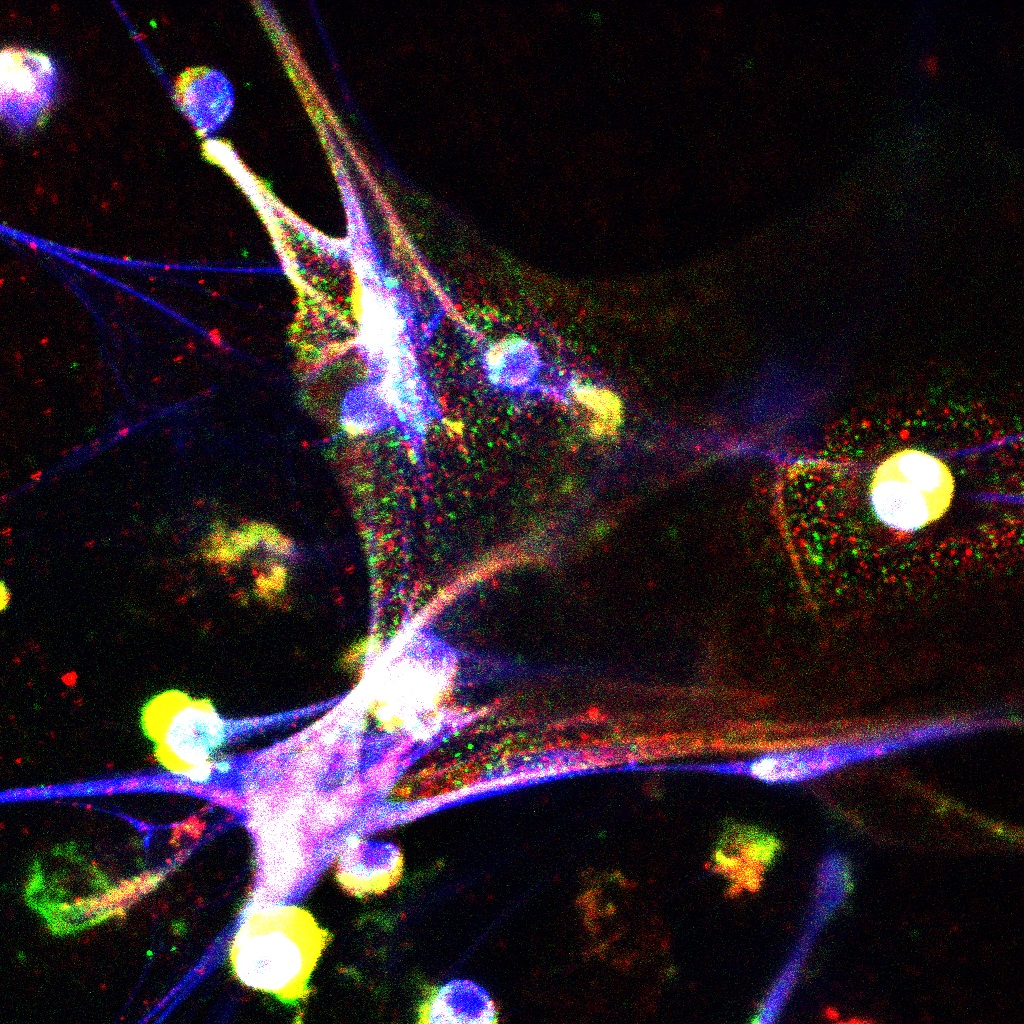
Image of a neutrophil extracellular trap
Publication:
Albrengues J, Shields MA, Ng D, et al. 2018. Neutrophil extracellular traps produced during inflammation awaken dormant cancer cells in mice. Science 361(6409).
Link:
Last updated Thursday, December 5, 2024














