Breast Cancer
Atf3 in Non-Cancer Host Cells is Linked to Chemotherapy-Enhanced Metastasis in Breast Cancer



Posted June 27, 2018
Tsonwin Hai, Ph.D, Ohio State University

Even though chemotherapy is typically effective at reducing the size of primary tumors in breast cancer patients, tumor relapse at the primary tumor site and/or distant tissues is common. Chemotherapeutic agents, such as paclitaxel (PTX), induce changes within both the tumor and host microenvironments, and are thought to contribute to increased chemoresistance and cancer progression. 1,2 Dr. Hai and her research team study the role of a stress-inducible gene, activating transcription factor 3 (Atf3), in breast cancer metastasis using mouse models. While other researchers, using cultured cells, showed that PTX increases the expression of Atf3 in cancer cells and caused cell death 3, Dr. Hai, using mouse models, showed that Atf3 expression in non-cancerous host cells augments breast cancer cell metastasis.4
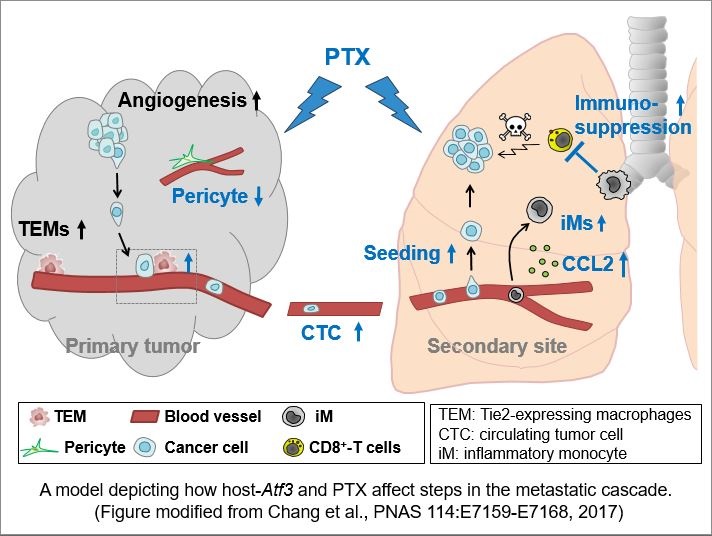
With support from a Fiscal Year 2013 Breast Cancer Research Program Breakthrough Award - Funding Level 2, Dr. Hai and her research team investigated the role of non-cancer cell host Atf3 in breast cancer metastasis in the presence or absence of PTX. In a recent paper published in Proceedings of the National Academy of Sciences, Dr. Hai used mouse models that express or lack Atf3 and metastatic breast cancer cell lines to show that increased breast cancer metastasis after treatment with PTX is dependent on Atf3 in non-cancer host cells. In addition, an increase was observed in the expression of proangiogenic genes, as well as microvascular densities, an indicator of angiogenesis, in tumors isolated from Atf3-expressing mice treated with PTX. Importantly, the number of circulating tumor cells (CTCs) was also found to be elevated in mice expressing Atf3 compared to mice lacking expression of Atf3. The number of CTCs was further increased with treatment of PTX in the Atf3-expressing mice supporting the hypothesis that non-cancer host cell Atf3 plays a role in enhanced breast cancer metastasis in the presence of PTX. Thus, despite reducing the tumor size, PTX changed the properties in the tumor and enable more cancer cells to escape.
To determine whether pretreatment with chemotherapy affected breast cancer lung colonization, Dr. Hai and her research team treated the mice with PTX prior to cancer cell inoculation. They reported that pre-treatment with PTX enhanced breast cancer lung colonization in the Atf3-expressing mice compared to mice lacking expression of Atf3. In addition, the research team found that increased lung metastases correlated with a decrease in cell-death markers and cytotoxic T-cells in the lungs of Atf3-expressing mice treated with PTX, further supporting the role of non-cancer host cell Atf3 in enhanced breast cancer metastasis following chemotherapy treatment.
In summary, they showed that chemotherapy - a stress signal - via inducing Atf3 alters immune responses that not only promote more cancer cells (the 'seeds') to escape from the primary tumors but also change the distant tissues (the 'soil') to be more hospitable to cancer cells (as diagrammed in the figure). The net result is that chemotherapy, despite reducing primary tumor size, paradoxically increases metastasis.
Using a publicly available genome-wide expression search engine, Oncomine, Dr. Hai looked to test the relevance of their observations from mouse models to human. Mining a patient database indicated that breast cancer patients who had undergone neoadjuvant chemotherapy treatment had increased expression of ATF3 in breast tumor stroma. Additionally, similar to what was observed in the mice, breast cancer patients with metastatic disease and ATF3 expression in tumor stroma also had increased expression of genes shown to create an immunosuppressive microenvironment, which is more hospitable to cancer cells.
Taken together, Dr. Hai and her research team have shown that not only is increased breast cancer metastasis dependent on non-cancer host cell Atf3 expression, but that Atf3 in the non-cancer host cells further drives breast cancer metastasis in the presence of PTX. These results suggest that targeting the effects of host cell ATF3 could help counteract the pro-metastatic effects of chemotherapy for breast cancer patients.
Publication:
Chang Y, Jalgainkar S, Middleton J, et al. 2017. Stress-inducible gene Atf3 in the noncancer host cells contributes to chemotherapy-exacerbated breast cancer metastasis. Proc Natl Acad Sci U S A 114(34):E7159-E7168.
References:
1 Ruffell, B. and Coussens, L.M. 2015. Macrophages and therapeutic resistance in cancer. Cancer Cell 27, 462-472.
2 Ran, S. The role of tlr4 in chemotherapy-driven metastasis. 2015. Cancer Res. 75, 2405-2410.
3 Oh YK, Lee HJ, Jeong MH, et al. 2008. Role of activating transcription factor 3 on Tap73 stability and apoptosis in paclitaxel-treated cervical cancer cells. Mol Cancer Res 6:1232-1249.
4 Wolford CC, McConoughey SJ, Jalgaonkar SP, et al. 2013. Transcription factor ATF3 links host adaptive response to breast cancer metastasis. J Clin Invest 123:2893-2906.
Last updated Wednesday, March 12, 2025














