Breast Cancer
Imaging Agent, Flurodeoxyglucose (18F-FDG), Transforms Chemotherapeutics into Phototherapy for Precise Treatment of Metastatic Breast Cancer



Posted October 3, 2018
Samuel Achilefu, Ph.D., Washington University School of Medicine

Washington University School
of Medicine
Chemotherapy is highly toxic to cancer patients, but also the last line of treatment for patients with lethal metastatic disease. In breast cancer, the bone marrow is one of the most common sites of metastasis. While the purpose of chemotherapy is to kill the cancer cells which are rapidly proliferating, bone marrow contains a variety of normal cells including pluripotent stem cells, which are highly sensitive to chemotherapy, resulting in toxic side effects not well tolerated by patients. As such, researchers are exploring second-generation chemotherapeutic agents that can treat metastatic lesions with higher specificity and lower toxicity. One such approach, phototherapy, utilizes light to selectively and spatially activate light-sensitive agents capable of killing tumor cells. However, phototherapy is limited to tumors that are easily accessible, because light does not penetrate deeply into tissues. Therefore, phototherapy has not been an option for treating tumors that metastasize to impenetrable tissues such as the bone marrow. As such, alternative approaches to phototherapy are being explored.
With support from a Fiscal Year 2015 Distinguished Investigator Award, Dr. Achilefu aimed to use Cerenkov radiation-induced therapy (CRIT), an alternative approach to phototherapy, to deliver and activate a light-sensitive agent directly to breast cancer metastatic bone lesions and selectively eliminate the tumor cells. CRIT is the process in which a light given off from a molecule (termed Cerenkov radiation [CR]) in conjunction with other radionuclide stimulation mechanisms, activates a light-sensitive agent, leading to the generation of reactive oxygen species (ROS), which can then enhance the activation of cell death pathways. CR is emitted by many U.S. Food and Drug Administration (FDA)-approved imaging agents used clinically to visualize biomolecules in patients. One such imaging agent, flurodeoxyglucose (18F-FDG), is widely used in clinical practice to aid clinicians with staging cancer and identifying metastatic foci. 18F-FDG specifically homes to rapidly dividing tumor cells, illuminating them, so that they can be visualized via positron emission tomography (PET) scans.
Recently published in Nature Communications, the results of Dr. Achilefu's study demonstrated that, by incorporating titanocene (TC), a light-sensitive chemotherapeutic agent, into human serum albumin (HSA) nanoparticles (HSA-TC), he was able to specifically home the agent to bone metastatic lesions and subsequently use the CR emitted by 18F-FDG to activate TC within tumor cells. Using a highly metastatic and aggressive breast cancer cell line, Dr. Achilefu stably introduced green fluorescent protein into the cells so that his team could visually monitor tumor cell response to CRIT in real time, in vivo. Mice injected with these cells developed metastatic lesions in the bone, lung, liver, and spine, similar to what is observed in the clinic with advanced metastatic breast cancer patients. Mice that received the CRIT treatment showed a decrease in overall metastatic burden, and metastatic tumors appeared to have halted growth.
The biodistribution of HSA-TC and potential bone marrow toxicity of CRIT were also examined by Dr. Achilefu and his team. HSA-TC concentration was highest 3 hours post-injection in mice harboring breast cancer bone metastases, particularly where the metastatic burden was highest. The concentration of HSA-TC decreased gradually over time so that by 24 hours, the body had completely cleared HSA-TC from its system. Importantly, analysis of bone marrow progenitor stem cells isolated from mice treated with CRIT revealed no adverse effects on the stem cells' ability to proliferate or be used for hematopoietic stem cell transplantation in mice. More importantly, bone marrow repopulation experiments demonstrated that there was no significant difference in hematopoietic reconstitution in mice that received untreated or CRIT-treated bone marrow progenitor stem cells, confirming the lack of off-target effects.
Although there is much work to still be performed, including a comprehensive understanding of the radionuclide stimulation mechanism, the results generated by Dr. Achilefu and his team signify a promising future for the treatment of metastatic cancer by using light-sensitive chemotherapeutic agents to specifically target metastatic lesions. If successful, phototherapy could serve as a new treatment option for advanced metastatic breast cancer patients.
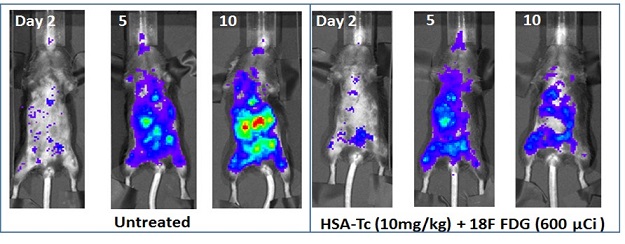
Bioluminescence imaging of in vivo radionuclide-stimulated therapy of highly aggressive metastatic PyMT-B01 tumor in C57BL6 mice. Untreated mice and those treated with a radionuclide alone (FDG) or a light-sensitive drug (TC) formulated in human serum albumin (HSA) dye rapidly within 10 days of the study compared to those treated with the stimulated therapy, which had lower tumor burden. Histologic analysis showed no adverse effects in non-tumor tissues.
Publication:
Kotagiri N, Cooper ML, Rettig M, et al. 2018. Radionuclides transform chemotherapeutics into phototherapeutics for precise treatment of disseminated cancer. Nature Communications 9(1):275.
Link:
Last updated Wednesday, March 12, 2025














