Breast Cancer
RON kinase: A Target for Treatment of Cancer-Induced Bone Destruction and Osteoporosis



Posted October 25, 2017
Alana Welm, Ph.D., University of Utah
Kelsi Andrade (formerly Kelsi Kretschmann), Ph.D., University of Oklahoma Health Science Center

Alana Welm, Ph.D., University of Utah

Kelsi Andrade, Ph.D., University of Oklahoma Health Science Center
Ultimately, more than 70% of metastatic breast cancer patients develop metastases in the bone. Bone metastases cause debilitating pain in patients as a result of nerve compression, hypercalcemia, and/or fractures. These complications are due to osteolysis, or bone loss, triggered by tumor cell interaction with bone tissue. Because the mechanisms leading to the establishment of bone metastases are poorly understood, the development of new therapeutics able to inhibit and treat bone metastasis has been hindered. In mouse models, host expression of macrophage stimulating protein (MSP) has been found to promote the metastasis of breast tumors to several sites including bone. Moreover, it was found that expression of MSP and its binding receptor, RON kinase, in human breast tumor samples was associated with poor prognosis.1 Therefore, with a Fiscal Year 2007 (FY07) Breast Cancer Research Program (BCRP) Era of Hope Scholar Award, Dr. Alana Welm sought to determine whether targeting of the MSP pathway via inhibition of RON kinase was a feasible therapeutic strategy for preventing metastatic osteolysis. Complementing Dr. Welm's award, Dr. Kelsi Andrade, a previous predoctoral trainee in Dr. Welm's group, received an FY09 BCRP Predoctoral Traineeship Award to elucidate the mechanisms underlying bone loss in MSP-expressing metastatic breast tumors and to determine how RON inhibitors nullify MSP activity.
To determine the osteolytic function of MSP/RON kinase signaling, Dr. Welm and her team used a genetically engineered mouse model that lacked the expression of the tyrosine kinase domain of RON kinase (RON TK-/-); rendering the receptor inactive2. As recently published in Science Translational Medicine, Dr. Welm was able to successfully show that inhibition of RON kinase function was both feasible and effective in reducing bone loss in mice that underwent intratibial injections with PyMT mouse mammary tumor cells engineered to express MSP. Results demonstrated that, when MSP-expressing breast cancer cells were injected into RON TK-/- or wild-type (RON TK+/+) animals, only the bones of mice lacking expression of functional RON kinase were protected from osteolysis. Similarly, treatment of wild-type animals with a RON kinase inhibitor, BMS-777607/ASLAN002, 3 days after tumor cell injection (pre-osteolysis), significantly inhibited MSP-induced bone loss. In a more clinically relevant setting (post-osteolysis), the team determined that BMS-777607/ASLAN002 was able to prevent further bone destruction when administered after osteolytic lesions could be detected by x-ray.
Dr. Welm also examined whether MSP/RON kinase signaling functions independently of the receptor activator of nuclear factor ?B ligand (RANKL) signaling; a well-known and key signaling pathway in promoting bone loss in osteolytic lesions. In vivo analyses showed that RON inhibition did not affect serum levels of RANKL, nor was inhibition of RANKL signaling with muRANK-Fc (mouse-specific RANKL antagonist) effective in preventing bone loss in MSP-expressing breast tumors. Moreover, Dr. Welm's team found that MSP-expressing breast tumors mediate bone loss through activation of osteoclasts (macrophage-like bone cells responsible for osteolytic lesions); whereas RANKL promotes osteoclast differentiation and activity, suggesting that MSP/RON kinase function in parallel with, but separate from, RANKL to promote metastatic bone destruction.
To test whether the in vivo findings were translatable into humans, Dr. Welm's team obtained plasma samples from a Phase I Clinical Trial (NCT01721148) of BMS-777607/ASLAN002 in metastatic cancer patients. Plasma samples from patients were tested for ß-cross-linked C telopeptide (CTX), a serum biomarker used to measure osteolysis, before and after treatment with BMS-777607/ASLAN002. Results demonstrated that 13 of 21 treated subjects (62%) exhibited decreased CTX levels after treatment with the RON kinase inhibitor. More importantly, the best treatment response was observed in women, with 9 of 11 (82%) demonstrating a decrease in CTX levels after treatment. The decrease in CTX was accompanied by a concomitant increase in bone-specific alkaline phosphatase (BSAP) levels, a biomarker of bone repair. BMS-777607/ASLAN002 was also found to be well tolerated by patients.
Current treatments for cancer patients with bone metastases consist of either bisphosphonates (i.e., Zoledronic acid) or RANKL inhibitors (i.e. Denosumab), which inhibit osteoclast activation or differentiation, respectively. Unfortunately, patients often become unresponsive to treatments and metastatic disease progresses, killing the patient. Dr. Welm and her team were able to elucidate a novel druggable pathway that functions separately from RANKL to promote bone loss in breast cancer patients. Because 40% of breast tumors overexpress MSP, this study paves the way for a new clinically relevant therapeutic that could be used in combination with bisphosphonates and RANKL inhibitors to treat a large proportion of metastatic cancer patients.
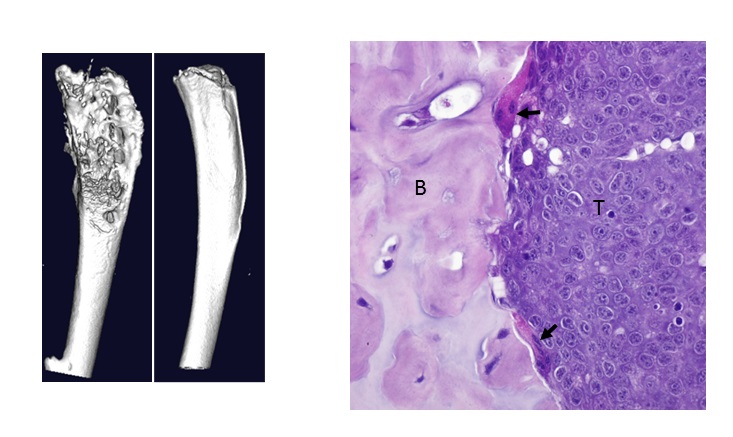
Left: MicroCT images demonstrating bone destruction in tumor-bearing tibia (left) compared to normal bone architecture in contralateral uninjected tibia (right) in a mouse model of bone metastasis.
Right: Bone-resorbing osteoclasts (arrows) at the interface of PyMT tumor cells (T) and bone (B) in mice.
Publication:
Andrade K, Fornetti J, Zhao L, Miller SC, Randall RL, Anderson N, Waltz SE, McHale M, and Welm AL. 2017. RON kinase: A target for treatment of cancer-induced bone destruction and osteoporosis. Sci Transl Med 9:1-11.
References:
1 Welm AL, Sneddon JB, Taylor C, Nuyten DS, van de Vigver MJ, Hasequawa BH, and Bishop JM. 2007. The macrophage-stimulating protein pathway promotes metastasis in a mouse model for breast cancer and predicts poor prognosis in humans. Proc Natl Acad Sci USA 104(18):7570-7575.
2 Waltz SE, Eaton L, Toney-Earley K, Hess KA, Peace BE, Ihlendorf JR, Wang MH, Kaestner KH, Degen SJ. 2001. Ron-mediated cytoplasmic signaling is dispensable for viability but is required to limit inflammatory responses. J Clin Invest. 108(4):567-76.
Links:
Public and Technical Abstracts: Attacking Breast Cancer: From the Lab to the Clinic
Public and Technical Abstracts: The Role of MSP Signaling in Metastasis of Breast Cancer to Bone

Kelsi Andrade, Ph.D., University of Oklahoma Health Science Center
Ultimately, more than 70% of metastatic breast cancer patients develop metastases in the bone. Bone metastases cause debilitating pain in patients as a result of nerve compression, hypercalcemia, and/or fractures. These complications are due to osteolysis, or bone loss, triggered by tumor cell interaction with bone tissue. Because the mechanisms leading to the establishment of bone metastases are poorly understood, the development of new therapeutics able to inhibit and treat bone metastasis has been hindered. In mouse models, host expression of macrophage stimulating protein (MSP) has been found to promote the metastasis of breast tumors to several sites including bone. Moreover, it was found that expression of MSP and its binding receptor, RON kinase, in human breast tumor samples was associated with poor prognosis.1 Therefore, with a Fiscal Year 2007 (FY07) Breast Cancer Research Program (BCRP) Era of Hope Scholar Award, Dr. Alana Welm sought to determine whether targeting of the MSP pathway via inhibition of RON kinase was a feasible therapeutic strategy for preventing metastatic osteolysis. Complementing Dr. Welm's award, Dr. Kelsi Andrade, a previous predoctoral trainee in Dr. Welm's group, received an FY09 BCRP Predoctoral Traineeship Award to elucidate the mechanisms underlying bone loss in MSP-expressing metastatic breast tumors and to determine how RON inhibitors nullify MSP activity.
To determine the osteolytic function of MSP/RON kinase signaling, Dr. Welm and her team used a genetically engineered mouse model that lacked the expression of the tyrosine kinase domain of RON kinase (RON TK-/-); rendering the receptor inactive2. As recently published in Science Translational Medicine, Dr. Welm was able to successfully show that inhibition of RON kinase function was both feasible and effective in reducing bone loss in mice that underwent intratibial injections with PyMT mouse mammary tumor cells engineered to express MSP. Results demonstrated that, when MSP-expressing breast cancer cells were injected into RON TK-/- or wild-type (RON TK+/+) animals, only the bones of mice lacking expression of functional RON kinase were protected from osteolysis. Similarly, treatment of wild-type animals with a RON kinase inhibitor, BMS-777607/ASLAN002, 3 days after tumor cell injection (pre-osteolysis), significantly inhibited MSP-induced bone loss. In a more clinically relevant setting (post-osteolysis), the team determined that BMS-777607/ASLAN002 was able to prevent further bone destruction when administered after osteolytic lesions could be detected by x-ray.
Dr. Welm also examined whether MSP/RON kinase signaling functions independently of the receptor activator of nuclear factor ?B ligand (RANKL) signaling; a well-known and key signaling pathway in promoting bone loss in osteolytic lesions. In vivo analyses showed that RON inhibition did not affect serum levels of RANKL, nor was inhibition of RANKL signaling with muRANK-Fc (mouse-specific RANKL antagonist) effective in preventing bone loss in MSP-expressing breast tumors. Moreover, Dr. Welm's team found that MSP-expressing breast tumors mediate bone loss through activation of osteoclasts (macrophage-like bone cells responsible for osteolytic lesions); whereas RANKL promotes osteoclast differentiation and activity, suggesting that MSP/RON kinase function in parallel with, but separate from, RANKL to promote metastatic bone destruction.
To test whether the in vivo findings were translatable into humans, Dr. Welm's team obtained plasma samples from a Phase I Clinical Trial (NCT01721148) of BMS-777607/ASLAN002 in metastatic cancer patients. Plasma samples from patients were tested for ß-cross-linked C telopeptide (CTX), a serum biomarker used to measure osteolysis, before and after treatment with BMS-777607/ASLAN002. Results demonstrated that 13 of 21 treated subjects (62%) exhibited decreased CTX levels after treatment with the RON kinase inhibitor. More importantly, the best treatment response was observed in women, with 9 of 11 (82%) demonstrating a decrease in CTX levels after treatment. The decrease in CTX was accompanied by a concomitant increase in bone-specific alkaline phosphatase (BSAP) levels, a biomarker of bone repair. BMS-777607/ASLAN002 was also found to be well tolerated by patients.
Current treatments for cancer patients with bone metastases consist of either bisphosphonates (i.e., Zoledronic acid) or RANKL inhibitors (i.e. Denosumab), which inhibit osteoclast activation or differentiation, respectively. Unfortunately, patients often become unresponsive to treatments and metastatic disease progresses, killing the patient. Dr. Welm and her team were able to elucidate a novel druggable pathway that functions separately from RANKL to promote bone loss in breast cancer patients. Because 40% of breast tumors overexpress MSP, this study paves the way for a new clinically relevant therapeutic that could be used in combination with bisphosphonates and RANKL inhibitors to treat a large proportion of metastatic cancer patients.

Left: MicroCT images demonstrating bone destruction in tumor-bearing tibia (left) compared to normal bone architecture in contralateral uninjected tibia (right) in a mouse model of bone metastasis.
Right: Bone-resorbing osteoclasts (arrows) at the interface of PyMT tumor cells (T) and bone (B) in mice.
Publication:
Andrade K, Fornetti J, Zhao L, Miller SC, Randall RL, Anderson N, Waltz SE, McHale M, and Welm AL. 2017. RON kinase: A target for treatment of cancer-induced bone destruction and osteoporosis. Sci Transl Med 9:1-11.
References:
1 Welm AL, Sneddon JB, Taylor C, Nuyten DS, van de Vigver MJ, Hasequawa BH, and Bishop JM. 2007. The macrophage-stimulating protein pathway promotes metastasis in a mouse model for breast cancer and predicts poor prognosis in humans. Proc Natl Acad Sci USA 104(18):7570-7575.
2 Waltz SE, Eaton L, Toney-Earley K, Hess KA, Peace BE, Ihlendorf JR, Wang MH, Kaestner KH, Degen SJ. 2001. Ron-mediated cytoplasmic signaling is dispensable for viability but is required to limit inflammatory responses. J Clin Invest. 108(4):567-76.
Links:
Public and Technical Abstracts: Attacking Breast Cancer: From the Lab to the Clinic
Public and Technical Abstracts: The Role of MSP Signaling in Metastasis of Breast Cancer to Bone
Last updated Wednesday, March 19, 2025














