Breast Cancer
RB1 Deficiency in Triple-Negative Breast Cancer Induces Mitochondrial Protein Translation



Posted November 30, 2016
Robert A. Jones, Ph.D., University Health Network, Toronto

Robert A. Jones, Ph.D., University Health Network, Toronto

Eldad Zacksenhaus, Ph.D., University Health Network, Toronto
Triple-negative breast cancer (TNBC) is defined as lacking expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER-2). Consequently, none of the targeted therapies developed for ERa+ and HER-2+ breast cancer have been found to be effective in the treatment of TNBC. Current therapeutic options for patients diagnosed with TNBC include surgery, radiation, and chemotherapy. Although TNBCs do initially respond to chemotherapy, resistance often develops, leaving few therapeutic options. Therefore, a better understanding of the underlying biology of TNBC is essential so that targets for therapeutic intervention can be identified and developed into more effective treatment modalities.
Tumor protein 53 (p53) and Retinoblastoma 1 (Rb1) are two tumor suppressor proteins that have been found to be frequently lost together in 28%-40% of TNBC cases. In TNBC, loss of p53 or Rb1 has been shown to correlate with tumor cells that display an aggressiveness that could be attributed to a higher proportion of tumor-initiating cancer stem cell (CSC)-like cells that have been found to drive relapse and resistance to chemotherapy. Because p53 and Rb1 are often lost during tumor progression, they are not directly targetable; however, their downstream effectors could be. With support from an FY09 BCRP Postdoctoral Fellowship Award, Dr. Robert Jones set out to elucidate vulnerabilities that could be therapeutically exploited downstream of p53 and Rb1 signaling.
In a landmark publication released this year in the Journal of Clinical Investigation, Dr. Jones, together with a large team of researchers working under the guidance of Dr. Eldad Zacksenhaus, were able to demonstrate for the first time that a combined loss of p53 and Rb1 (p53/Rb1-deficient) elevated levels of genes associated with the mitochondrial protein translation (MPT) pathway which were susceptible to the FDA-approved MPT inhibitor tigecycline (TIG). TIG was identified as the most effective drug against p53/Rb1-deficient TNBC cell lines during a repurposing drug screen of 312 U.S. Food and Drug Administration (FDA)-approved drugs. Results indicated that p53/Rb1-deficient TNBC cell lines were significantly more sensitive to TIG when compared to p53-mutant/Rb1-proficient cell lines. Additionally, analyses revealed that p53/Rb1-deficient TNBC cells displayed reduced cellular proliferation and mitochondrial protein expression upon TIG treatment, suggesting that p53/Rb1-deficient TNBC cells are dependent on MPT. Moreover, in an Rb1-dependent manner, the tumor-initiating CSC-like fraction could also be reduced with TIG treatment. Conversely, Rb1-proficient TNBC cell lines showed an enhancement in the CSC fraction upon TIG treatment. An increase in the tumor-initiating CSC-like cellular population, as seen in Rb1-proficient TNBC cell lines, could result in relapse and resistance to chemotherapy, which would be detrimental to patients; therefore these results suggest patient stratification for treatment would be essential.
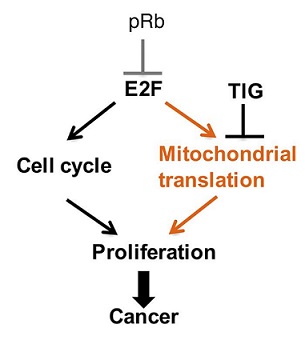
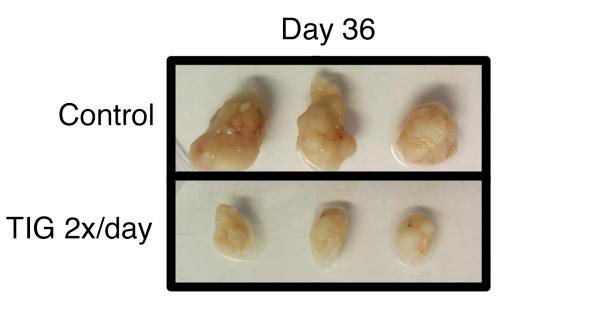
This work highlights the importance of patient stratification prior to treatment and demonstrates that tumor vulnerabilities can be identified within TNBC tumors, and, more importantly, these vulnerabilities can be exploited therapeutically. Dr. Jones and colleagues' research supports not only a role for TIG and other MPT inhibitors in the treatment of p53/Rb1-deficient TNBC but also provides insight into the biology of these aggressive tumors that could be exploited to develop other novel targeted therapeutics.
Publication:
Jones RA, Robinson TJ, Liu JC, et al. 2016. RB1 deficiency in triple-negative breast cancer induces mitochondrial protein translation. J Clin Invest pii:81568 [Epub ahead of print]
Link:
Last updated Wednesday, March 12, 2025














