


Posted August 19, 2015
Jing Yang and Adam Engler, University of California, San Diego
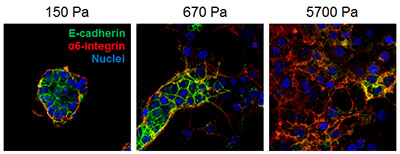
 The presence of an area of increased stiffness within a breast tumor, or a fibrotic focus, is associated with cancer progression, distant metastases, and overall poor outcome. Also associated with metastases and poor outcome is an enhanced "stemness" of breast cancer cells. Cancer stem cells adopt properties making them better able to migrate, invade, survive in the circulation, and colonize in foreign tissue. While the underlying mechanism for why tissue, or matrix, stiffness promotes cancer progression is not fully understood, Drs. Yang and Engler have hypothesized that tissue rigidity regulates breast cancer stem cell properties and function, thereby promoting tumor development and chemoresistance. With funding from an FY12 BCRP Idea Award and Breast Cancer Stamp funds, Drs. Yang and Engler have found that increasing matrix stiffness promotes epithelial-to-mesenchymal transition (EMT) in human breast cancer cells and that this regulation depends on the adhesion receptor ß1 integrin and the transcription factor TWIST1. Their findings, published in a 2015 Nature Cell Biology article, suggest a newly identified paradigm in which increasing matrix stiffness induces an integrin-dependent cell signaling cascade and subsequent release of TWIST1 from its cytoplasmic sequestration factor G3BP2. TWIST1 is then able to enter the nucleus and induce the transcription of EMT and invasion-promoting factors. Depletion of G3BP2 promoted breast cancer invasion and metastasis in mice, and reduced expression of G3BP2 in combination with increased matrix stiffness predicts poor survival in breast cancer patients. Cells that have undergone EMT are shown to present many properties of cancer stem cells, thus the ß1-G3BP2-TWIST1 mechanotransduction pathway may be a key to the regulation of breast cancer stem cell properties and chemoresistance. Further investigation into this pathway may lead to novel therapeutic strategies that will be able to halt progression of a tumor before it has the chance to metastasize. Furthermore, this project highlights the effectiveness of the coming together of two labs with complementary sets of expertise, breast cancer biology (the Yang lab), and bioengineering and stem cell biology (the Engler lab). The collaboration has proved to be essential to implement this research program.
The presence of an area of increased stiffness within a breast tumor, or a fibrotic focus, is associated with cancer progression, distant metastases, and overall poor outcome. Also associated with metastases and poor outcome is an enhanced "stemness" of breast cancer cells. Cancer stem cells adopt properties making them better able to migrate, invade, survive in the circulation, and colonize in foreign tissue. While the underlying mechanism for why tissue, or matrix, stiffness promotes cancer progression is not fully understood, Drs. Yang and Engler have hypothesized that tissue rigidity regulates breast cancer stem cell properties and function, thereby promoting tumor development and chemoresistance. With funding from an FY12 BCRP Idea Award and Breast Cancer Stamp funds, Drs. Yang and Engler have found that increasing matrix stiffness promotes epithelial-to-mesenchymal transition (EMT) in human breast cancer cells and that this regulation depends on the adhesion receptor ß1 integrin and the transcription factor TWIST1. Their findings, published in a 2015 Nature Cell Biology article, suggest a newly identified paradigm in which increasing matrix stiffness induces an integrin-dependent cell signaling cascade and subsequent release of TWIST1 from its cytoplasmic sequestration factor G3BP2. TWIST1 is then able to enter the nucleus and induce the transcription of EMT and invasion-promoting factors. Depletion of G3BP2 promoted breast cancer invasion and metastasis in mice, and reduced expression of G3BP2 in combination with increased matrix stiffness predicts poor survival in breast cancer patients. Cells that have undergone EMT are shown to present many properties of cancer stem cells, thus the ß1-G3BP2-TWIST1 mechanotransduction pathway may be a key to the regulation of breast cancer stem cell properties and chemoresistance. Further investigation into this pathway may lead to novel therapeutic strategies that will be able to halt progression of a tumor before it has the chance to metastasize. Furthermore, this project highlights the effectiveness of the coming together of two labs with complementary sets of expertise, breast cancer biology (the Yang lab), and bioengineering and stem cell biology (the Engler lab). The collaboration has proved to be essential to implement this research program.
Publications:
Wei S, Fattet L, Tsai J, Guo Y, Pai V, Majeski H, Chen A, Sah R, Taylor S, Engler A, and Yang J. 2015. Matrix stiffness drives epithelial-mesenchymal transition and tumour metastasis through TWIST1-G3BP2 mechanotransduction pathway. Nature Cell Biology 17(5): 678-688.
Haeyun Jung, Laurent Fattet, and Jing Yang. 2014 Molecular Pathways: Linking Tumor Microenvironment to Epithelial-Mesenchymal Transition in Metastasis. Clinical Cancer research 21(5); 1-7.
Tsai J and Yang J. 2013. Epithelial-mesenchymal plasticity in carcinoma metastasis. Genes and Development 27(20): 2192-2206.
Links:
Public and Technical Abstracts: Regulation of Breast Cancer Stem Cell by Tissue Rigidity














