


Posted December 4, 2014
Ben K. Seon, Ph.D., Roswell Park Cancer Institute, Buffalo, New York
 A fundamental property of cancer is the formation of new blood vessels that transport essential nutrients for growth to tumors, a process known as angiogenesis. Because this process is important for tumor development and is required for tumor metastasis, recent therapeutic efforts have focused on targeting important regulators of angiogenesis. The most widely tested anti-angiogenic therapies have focused on targeting vascular endothelial growth factor (VEGF), a protein produced by cells that stimulates angiogenesis. Some drugs that inhibit VEGF, such as bevacizumab (Avastin®), have shown clinical promise, but have shown limited therapeutic efficacy and significant toxicity in breast cancer patients. Therefore, it is crucial to pursue other efforts to target tumor-associated angiogenesis with safe, effective therapeutics.
A fundamental property of cancer is the formation of new blood vessels that transport essential nutrients for growth to tumors, a process known as angiogenesis. Because this process is important for tumor development and is required for tumor metastasis, recent therapeutic efforts have focused on targeting important regulators of angiogenesis. The most widely tested anti-angiogenic therapies have focused on targeting vascular endothelial growth factor (VEGF), a protein produced by cells that stimulates angiogenesis. Some drugs that inhibit VEGF, such as bevacizumab (Avastin®), have shown clinical promise, but have shown limited therapeutic efficacy and significant toxicity in breast cancer patients. Therefore, it is crucial to pursue other efforts to target tumor-associated angiogenesis with safe, effective therapeutics.
Dr. Ben Seon has been at the forefront of developing an anti-angiogenic therapy that targets endoglin (ENG), a proliferation-associated protein that is strongly expressed in tumor-associated blood vessels while only very weakly expressed in normal blood vessels. He and his research team, with support from an FY96 Breast Cancer Research Program (BCRP) Research with Translational Potential Award, have developed anti-ENG monoclonal antibodies (mAbs) that not only inhibit metastasis by inhibiting angiogenesis, but also cause regression of established tumors by targeting its vasculature. The ENG-targeted breast cancer therapy can be applied to most of the heterogeneous groups of breast cancer that include the triple-negative breast cancer, estrogen receptor (ER)-positive/progesterone receptor (PR)-positive breast cancer and HER2-overexpressing breast cancer. An FY02 BCRP Clinical Translational Research Award provided Dr. Seon the funding to develop a novel human/mouse chimeric anti-ENG mAb called c-SN6j (also known as TRC105) that showed a manageable safety profile and significant clinical activity in a phase I clinical trial in which 50 patients with various advanced solid tumors including metastatic breast cancer were treated with TRC105.
With BCRP support and in partnership with Tracon Pharmaceuticals, Dr. Seon and colleagues initiated a phase 1B dose-finding study of TRC105 in combination with capecitabine for advanced/metastatic HER2-negative breast cancer patients and colorectal cancer patients. A total of 16 patients with advanced/metastatic solid tumors (12 breast and 4 colorectal cancer) were treated at two different TRC105 dose levels (4 patients at 7.5 mg/kg and 12 patients at 10 mg/kg) in this trial. Adverse events characteristic of individual drugs were not increased in frequency or severity when the two drugs were administered together. A heavily pretreated HER2-negative breast cancer patient remained on study for 9 months with a RECIST-defined partial response with 52% tumor reduction. Two additional metastatic HER2-negative breast cancer patients responded to the therapy for more than 5 months while a metastatic colorectal cancer patient responded to the therapy for more than 4 months.
Although this result is encouraging, Dr Seon hypothesizes that the therapeutic efficacy of combination therapy with TRC105 can be substantially improved by selection of better partnering drugs. To facilitate this drug selection, with BCRP support Dr. Seon and his team developed a novel genetically engineered mouse model (GEMM) expressing humanized ENG. This GEMM should serve as a valuable research tool for optimizing anti-ENG therapy.
In summary, BCRP support enabled Dr. Seon and his team to develop TRC105 for treating breast cancer as well as several other cancers. Fourteen clinical trials of TRC105 are listed on clinicaltrials.gov as of October 24, 2014. In addition to breast cancer trials, two of these trials are National Cancer Institute-sponsored large trials to treat recurrent glioblastoma multiforme and metastatic kidney cancer, respectively. Dr. Seon and his team are exerting a major effort to optimize the ENG-targeted breast cancer therapy. Encouraging results from Dr. Seon's work and these clinical trials may advance TRC105 as a new treatment for metastatic breast cancer and other solid tumors.
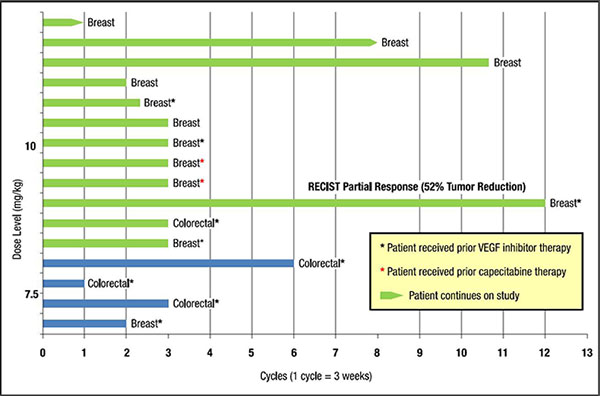
A phase 1B dose-escalation study of TRC105 in combination with capecitabine for patients with progressive or recurrent HER2-negative metastatic breast cancer and metastatic colorectal cancer. Dose escalation proceeded from 7.5 mg/kg (n = 4) to 10 mg/kg (n = 12) of TRC105 without development of dose-limiting toxicity. Twelve breast cancer patients and 4 colorectal cancer patients were treated in the presented figure. Ordinate: TRC dose level (mg/kg). Abscissa: Drug dosing cycle (1 cycle = 3 weeks).
Publications:
Toi H, Tsujie M, Haruta Y, et al. 2014. Facilitation of endoglin-targeting cancer therapy by development/utilization of a novel genetically engineered mouse model expressing humanized endoglin (CD105). Int J Cancer. doi: 10.1002/ijc 28994 [Epub ahead of print]
Rosen LS, Hurwitz HI, Wong MK, et al. 2012. A phase I first-in-human study of TRC105 (anti-endoglin antibody) in patients with advanced cancer. Clin Cancer Res 18(17):4820-4829.
Uneda S, Toi H, Tsujie T, et al. 2009. Anti-endoglin monoclonal antibodies are effective for suppressing metastasis and the primary tumors by targeting tumor vasculature. Int J Cancer 125(6):1446-1453.
Links:
Public and Technical Abstracts: Vascular-Targeting Anti-Angiogenic Therapy of Human Breast Cancer














