


Posted March 27, 2014
Holbrook E. Kohrt, M.D., Stanford University
 Despite the efficacy of monoclonal antibodies in the treatment of cancer, they have failed to be the "magic bullet" once hoped for. Most approaches to improve their anti-cancer activity, such as combination with chemotherapy, increase their toxicity and reduce the patient's quality of life. With support from an FY10 BCRP Postdoctoral Fellowship Award, Dr. Holbrook Kohrt identified a novel method of enhancing a patient's immune response to tumor-targeting monoclonal antibodies. By first targeting the tumor with an antibody to recruit the immune system, then following with an antibody that enhances the immune response, Dr. Kohrt developed a two-antibody approach that improves activity without increasing toxicity. Combination therapy for HER2-expressing breast cancer could eliminate the need for chemotherapy, thus preserving patient quality and longevity of life.
Despite the efficacy of monoclonal antibodies in the treatment of cancer, they have failed to be the "magic bullet" once hoped for. Most approaches to improve their anti-cancer activity, such as combination with chemotherapy, increase their toxicity and reduce the patient's quality of life. With support from an FY10 BCRP Postdoctoral Fellowship Award, Dr. Holbrook Kohrt identified a novel method of enhancing a patient's immune response to tumor-targeting monoclonal antibodies. By first targeting the tumor with an antibody to recruit the immune system, then following with an antibody that enhances the immune response, Dr. Kohrt developed a two-antibody approach that improves activity without increasing toxicity. Combination therapy for HER2-expressing breast cancer could eliminate the need for chemotherapy, thus preserving patient quality and longevity of life.
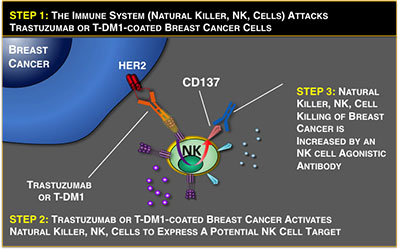
One in four to five women with breast cancer have tumors that express high levels of human epithelial growth factor receptor 2 (HER2). While HER2 signaling is involved in normal cell growth, increased HER2 signaling can lead to uncontrolled growth and the development of aggressive breast cancer. Trastuzumab is a monoclonal antibody (mAb) that binds the HER2 receptor on the surface of tumor cells and blocks downstream signaling pathways. In 2006 trastuzumab was approved to treat HER2-positive breast cancers. Despite the promising activity of trastuzumab, the response rates among patients with either refractory or advanced breast cancer remain suboptimal at less than 10%. Efforts to enhance the activity of trastuzumab have focused on various combinations with cytotoxic chemotherapy. This largely ignores and may partially antagonize the immunologic mechanism by which monoclonal antibodies function. One of the primary mechanisms of antitumor action is through antibody-dependent cell-mediated cytotoxicity whereby a natural killer (NK) cell binds to the antibody-targeted tumor cell and mediates the killing function. Therefore, therapies that augment NK cell function may uniquely offer the ability to improve the activity of monoclonal antibodies without increasing toxicity to non-cancer cells.
Dr. Kohrt sought to enhance trastuzumab's antitumor activity by increasing its ability to recruit the immune system to attack cancer cells. To do this, Dr. Kohrt tested the activity of fully competent NK cells. Following injection of trastuzumab, the NK cells that encountered trastuzumab-coated, HER2-expressing cells became activated and expressed the costimulatory receptor, CD137. When an agonist antibody targeting CD137 was added subsequent to trastuzumab injection a 50% increase in the cytotoxic activity of the NK cells was found. When the treatment regimen was repeated weekly for three weeks, the combination treatment resulted in prolonged control of tumor growth and a significant improvement in survival in a xenotransplant mouse model. Dr. Kohrt and colleagues further found that the success of this targeted antibody therapy, augmented with CD137, was not limited to HER2-positive breast cancers. They showed that the antitumor toxicity induced by rituximab, an antibody targeted to the immune system B cell protein, was also amplified by subsequent CD137 administration in an animal model of lymphoma.
Overall, the results of these studies demonstrated that sequential targeting of the CD137 receptor successfully stimulates the immune response and enhances antibody-dependent cell-mediated cytotoxicity. This novel strategy targeting both the tumor and the immune system may thus offer new opportunities to enhance the efficacy of therapeutic antibodies in breast cancer. Clinical investigations are now warranted to determine if this hypothesis holds true in patients.
Publication:
Kohrt HE, Houot R, Weiskopf K, Goldstein MJ, Scheeren F, Czerwinski D, Colevas AD, Weng WK, Clarke MF, Carlson RW, Stockdale FE, Mollick JA, Chen L, and Levy R. 2012. Stimulation of natural killer cells with a CD137-specific antibody enhnances trastuzumab efficacy in xenotransplant models of breast cancer. J Clin Invest 122(3):1066-1075.
Links:














