


Posted October 22, 2013
Erkki Ruoslahti, M.D., Ph.D., Sanford-Burnham Medical Research Institute, La Jolla
Shiladitya Sengupta, Ph.D., Massachusetts Institute of Technology
Roger Tsien, Ph.D., University of California, San Diego
 FY12 BCRP Collaborative Innovators Award
FY12 BCRP Collaborative Innovators Award
Chemotherapeutic drugs are a vital part of the frontline attack on cancer. But, while many patients have been successfully treated with drugs, the benefits of chemotherapy are tempered by its significant drawbacks. In addition to cancer cells, chemotherapies also kill some normal cells, such as those found in the bone marrow, hair, and the lining of the digestive tract. The ideal treatment would be a selective drug that targets tumor cells and does not affect normal, healthy cells.
The Breast Cancer Research Program's Collaborative Innovators Award was created to bring together scientists across different fields of expertise to come up with new and innovative ways to treat breast cancer. In fiscal year 2008, one such award went to a group of experts that included Dr. Erkki Ruoslahti, a cancer researcher from Sanford-Burnham Medical Research Institute in La Jolla, California, who specializes in blood vessel-based therapies, Dr. Shiladitya Sengupta, a nano-scale engineering expert from Harvard's Brigham and Women's Hospital, and Dr. Roger Tsien, a professor of pharmacology at the University of California, San Diego, School of Medicine and winner of the 2008 Nobel Prize in chemistry. This highly accomplished, interdisciplinary team of researchers proposed to use nanotechnology to make various "smart" chemotherapies that do more than serve as passive drug carriers. Their approaches employ nanomedicine and the unique expression of several molecules found inside tumors or tumor blood vessels. The research team has published a number of studies that demonstrate the promise of smart chemotherapies.
Cisplatin is a chemotherapeutic agent commonly used to treat malignancies in many types of cancers. Its use, however, is limited due to its severe toxicity, primarily to the kidney. Significant research effort has been focused on finding ways to boost cisplatin's destructive effects on tumors while limiting its effects on normal tissue. By taking advantage of the way cisplatin reacts with a new polymer material, Dr. Sengupta and his team created a cisplatin-polymer complex that assembles to form a nanoparticle that specifically enters cancer cells before the cisplatin drug is released. When tests were conducted in a breast cancer model, the platinum-bound nanoparticles were more effective at delaying tumor growth than cisplatin alone. Furthermore, systemic and kidney toxicities were also reduced with the nanoparticles. Drs. Sengupta and Ruoslahti have now made an advanced version of these nanoparticles by attaching iRGD to them for targeting to tumors. This work could one day lead to cisplatin treatments in which higher doses are both more effective and safer for breast cancer patients.
Another major limitation to chemotherapeutic treatment is the physical nature of tumors, which prevents the penetration of drugs into tumor tissue. To improve the potency of drugs that reach tumor cells, researchers have been pursuing ways to increase the amount of drug internalized by the cells once it reaches its destination. Dr. Ruoslahti's lab created a peptide, called iRGD, containing two sequences. One sequence binds integrin receptors on the surface of tumor cells and the endothelial cells of the tumor vasculature, while the other sequence promotes the peptide's entry into the tumor cell. In recent work, a tumor-homing signal, called the NGR sequence, was added to the peptide. The NGR sequence binds to the CD13 protein that is expressed in the endothelial cells of tumors. This smart tumor-homing and tissue-penetrating molecule, called iNGR, triggers the penetration of other molecules injected into the blood stream along with it. When the chemotherapeutic agent doxorubicin was administered simultaneously with iNGR in a mouse breast cancer model, higher amounts of doxorubicin accumulated within tumors, demonstrating greater effectiveness in slowing tumor growth than doxorubicin treatment alone. Importantly, enhancing doxorubicin's effectiveness occurred without increased side effects. Because it is not attached to the drug, iNGR could potentially be used to enhance the efficacy of other chemotherapeutics. Additionally, the sequences on iRGD and iNGR that give it its tumor-targeting and tissue-penetrating capabilities could be attached to other molecules to turn a passive delivery system into a smart one.
The ability of a surgeon to remove a tumor depends on his or her ability to differentiate tumor tissue from surrounding normal tissue. Dr. Tsien, recipient of the Nobel Prize for the discovery and development of green fluorescent protein (GFP), used extensively in medical research imaging, is now applying his expertise to improving the detection and treatment of cancer. Dr. Tsien's lab previously created "activatable cell-penetrating peptides" (ACPPs), polyarginine-based molecules that enter tumor cells much more than they enter normal cells. In work published in 2010, Dr. Tsien's lab attached the ACPPs with a fluorescent label and used them to guide surgery in a mouse model for breast cancer. The mice injected with ACPP showed a fivefold increase in tumor-free, long-term survival as compared with standard surgery. His team recently showed that even better results can be obtained by attaching two fluorescent labels simultaneously to the same ACPPs such that they respond in opposite directions when taken into tumors. Dr. Tsien's work with ACPPs shows that the value of nanoparticles isn't limited to their ability to kill tumor cells directly-they can also be used to improve methods that are already in practice.
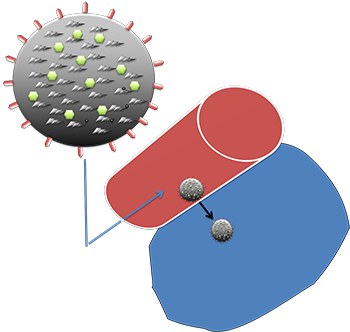
A nanoparticle that carries two kinds of payload, a drug and an imaging agent. Such nanoparticles are known as "theranostic" because of their ability to deliver therapy and serve diagnostic functions at the same time. The outer surface of the nanoparticle is coated with a peptide (spikes on the sphere) that binds to a receptor on the vascular wall of tumor blood vessels (but not in normal vessels). The homing peptides cause the nanoparticles to burrow through the vascular wall and into the tumor tissue, resulting in selective accumulation of the nanoparticles in the tumor (blue), which increase the activity of the enclosed drug and enhances the contrast from the imaging agent.
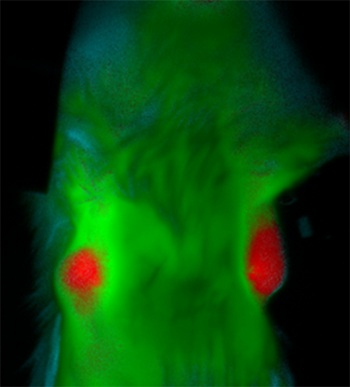
A ratiometric ACPP highlights orthotopic breast tumors in an immunocompetent mouse ~45 min after intravenous injection. Red color indicates the cleaved probe that clearly marks tumor, green color indicates the uncleaved probe in healthy tissue.
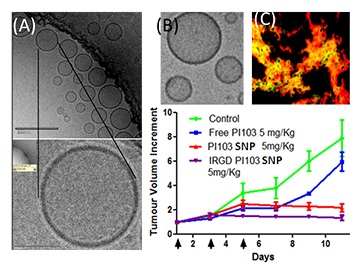
Various supramolecular nanoparticles developed in the Sengupta laboratory. (A) A Cryo-TEM shows molecularly-modified cisplatin analogues that have self-assembled into a supramolecular nanoparticle. (B) The chemistry was extended to a phosphatidylinositol 3 kinase inhibitor, which forms similar nanostructures. (C) When these nanoparticles were surface-coated with iRGD peptides developed in the Ruoslahti lab, the tumor gets lighted up with enhanced delivery of these nanoparticles specifically to the tumor. This results in increased efficacy as seen in the graph with these supramolecular nanoparticles (SNPs) in terms of breast tumor growth inhibition
Publications:
Agemy L, Kotamraju VR, Friedmann-Morvinski D, Sharma S, Sugahara KN, Ruoslahti E. 2013. Proapoptotic peptide-mediated cancer therapy targeted to cell surface p32. Mol Ther [Epub ahead of print].
Alberici L, Roth L, Sugahara KN, Agemy L, Kotamraju VR, Teesalu T, Bordignon C, Traversari C, Rizzardi GP, and Ruoslahti E. 2013. De novo design of a tumor-penetrating peptide. Cancer Res 73:804-812.
Savariar EN, Felsen C, Nashi N, Jiang T, Ellies LG, Steinbach P, Tsien RY, and Nguyen QT. 2013. Real time in vivo molecular detection of primary tumors and metastases with ratiometric activatable cell penetrating peptides. Cancer Res 73:855-864.
Nguyen QT, Olson ES, Aquilera TA, Jiang T, Scadeng M, Ellies LG, and Tsien RY. 2010. Surgery with molecular fluorescence imaging using activatable cell-penetrating peptides decreases residual cancer and improves survival. Proc Natl Acad Sci U S A 107:4317-4322.
Paraskar AS, Soni S, Chin KT, Chaudhuri P, Muto KW, Berkowitz J, Handlogten MW, Alves NJ, Bilgicer B, Dinulescu DM, Mashelkar RA, and Sengupta S. 2010. Harnessing structure-activity relationship to engineer a cisplatin nanoparticle for enhanced antitumor efficacy. Proc Natl Acad Sci U S A 107:12435-12440.
Links:
Abstracts for Dr. Ruoslahti
Public and Technical Abstracts: Hybrid Nanotechnologies for Detection and Synergistic Therapies for Breast Cancer
Abstracts for Dr. Tsien
Public and Technical Abstracts: Hybrid Nanotechnologies for Detection and Synergistic Therapies for Breast Cancer
Abstracts for Dr. Sengupta
Public and Technical Abstracts: Hybrid Nanotechnologies for Detection and Synergistic Therapies for Breast Cancer














