


Posted July 25, 2013
Heide L. Ford, Ph.D., University of Colorado Health Sciences Center
 The Breast Cancer Research Program (BCRP) aims to support innovative, high-impact research that will lead to solutions that will end breast cancer. This vision requires, not only supporting talented scientists who have already dedicated their lives to curing breast cancer, but also ensuring that the next generation of researchers are given the best opportunities to develop their talents. Dr. Heide Ford at the University of Colorado School of Medicine is a BCRP researcher whose commitment to pursuing innovative breast cancer research, as well as mentoring talented and successful graduate students and postdoctoral fellows, has revealed potentially groundbreaking results.
The Breast Cancer Research Program (BCRP) aims to support innovative, high-impact research that will lead to solutions that will end breast cancer. This vision requires, not only supporting talented scientists who have already dedicated their lives to curing breast cancer, but also ensuring that the next generation of researchers are given the best opportunities to develop their talents. Dr. Heide Ford at the University of Colorado School of Medicine is a BCRP researcher whose commitment to pursuing innovative breast cancer research, as well as mentoring talented and successful graduate students and postdoctoral fellows, has revealed potentially groundbreaking results.
Despite progress in diagnostic and therapeutic tools for breast cancers, tens of thousands of patients continue to die each year, mainly due to metastases. Uncovering the molecular basis of breast cancer metastases thus remains among the most important research priorities. Another priority is to not only develop effective therapies, but to develop targeted therapies that are less toxic than the limited options currently available.
To meet these challenges, Dr. Ford and colleagues have explored the possibility that proteins that play a crucial role in embryonic development, when regulated abnormally in adults, lead to cancer. They observed that many developmental processes, such as cell proliferation and cell survival, neovascularization, and tissue invasion also occur during tumorigenesis and metastasis. Their work centers on the Six1 protein, a member of the homeoprotein family of transcription factors that control embryonic development. Dr. Ford hypothesized that, even after functioning normally early in life, abnormal activation of Six1 in an adult could contribute to the formation of breast cancers. Importantly, while abundant during embryonic development, Six1 is not expressed in most adult tissues, minimizing its toxicity potential as a drug target.
 When Good Proteins Go Bad
When Good Proteins Go Bad
Dr. Ford received a BCRP Synergistic Idea Award for her study: "Structural and Functional Analyses of the Six1 Transcriptional Complex for Anti-Breast Cancer Drug Design" in fiscal year 2008. The Synergistic Idea Award was created to foster new collaborations to take on high-risk - and potentially high-reward - research challenges.
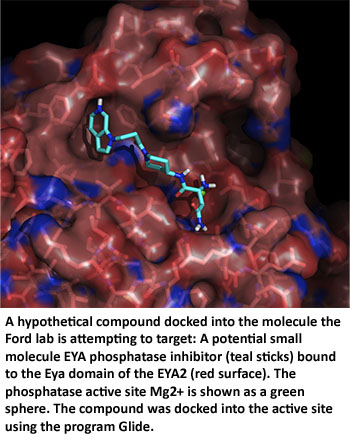
Dr. Ford, a breast cancer biologist, teamed up with Dr. Rui Zhao, a structural biologist and biochemist also at the University of Colorado, Denver. Working together, the labs showed that Six1 and its co-activator, Eya, form a transcriptional complex that is critical for both tumor growth and metastasis. They determined the crystal structure of the complex and identified inhibitors that are currently being tested in preclinical models. If the inhibitors are shown to work, the group hopes to begin testing them in clinical trials.
Six1 had never been identified as a potential clinical target for breast cancer prior to Dr. Ford's work. Through the Synergistic Idea Award, the discovery of the Six1/Eya complex's role in breast cancer has allowed the lab to explore further its potential as a therapeutic target. As Dr. Ford says:
"Funding through the DoD has been critical in allowing us to move this work forward, as it is high risk, but potentially very high gain. Drug development projects (particularly with developing novel inhibitors against new cancer targets) are typically very hard to get funded, and thus the funds received through the DoD to both understand metastasis and drug resistance, and to develop new inhibitors targeting the metastatic process, have been critical for our success to date and will hopefully allow us to test new ways to target metastasis."
Paving the Way for Future Research
Several Ford lab trainees have been awarded BCRP funding and are advancing in their career paths to become independent breast cancer researchers.
 Doug Micalizzi, M.D., Ph.D.
Doug Micalizzi, M.D., Ph.D.
Much of the foundation for the Synergistic Idea Award was laid down by a Ph.D. student in the Ford lab, Dr. Doug Micalizzi, who showed that Six1 overexpression induced metastasis. Supported by a predoctoral BCRP award in fiscal year 2006, Dr. Micalizzi showed that the actions of Six1 were most likely through reactivation of a developmental program called Epithelial-Mesenchymal Transition (EMT). He also showed that Six1 acts through the TGF-ß pathway.
"This award allowed me to pursue my Ph.D. thesis work without concern for my position in the lab. It gave me more freedom in my work. Since this award, I have earned my Ph.D., which was then followed by my M.D." Dr. Micalizzi's contributions to breast cancer research are far from over. He is currently a resident at Massachusetts General Hospital, after which he plans to pursue an oncology fellowship and "ultimately a career in academic medicine."
 Erica Koustas, Ph.D.
Erica Koustas, Ph.D.
Dr. Erica Koustas was awarded a predoctoral BCRP award in fiscal year 2006 to confirm the tumorigenic effects of Six1 in a mouse model of breast cancer. She not only confirmed that Six1 caused tumors in vivo, but subsequent analysis suggested that the cancer-promoting effects of Six1 may occur through disregulation of stem cells.
After completing her Ph.D., Dr. Koustas entered the world of science policy by winning a Science and Technology Policy Fellowship at the American Association for the Advancement of Science. Dr. Koustas feels her transition to science policy was a natural progression along a trajectory initiated with the BCRP award: "The BCRP award opened my eyes to the application of my research and its impact on breast cancer patients. I think a career in science policy was a natural next step for me."
 Susan Farabaugh, Ph.D.
Susan Farabaugh, Ph.D.
While a Ph.D. student in the Ford lab, Dr. Susan Farabaugh's research uncovered a molecule that showed promise as a therapeutic target with limited side effects. After receiving a predoctoral BCRP Award in fiscal year 2007, Dr. Farabaugh showed that the metastasis-promoting functions of Six1 were dependent on a co-factor, Eya2. Six1 and Eya2 function together in a transcriptional regulatory network during development, but the finding that Six1 is dependent on Eya2 to mediate its tumorigenic/metastatic effects presented a way to target the Six/Eya complex, as Six1 itself, as a DNA binding protein, was considered undruggable. Importantly Eya2 is abundant in breast cancers and even more so in metastatic tumors.
"At the time of publication, this research was the only one which examined the essential relationship of Six1 and Eya together in cancer. This relationship is imperative as the enzymatic activity of Eya lends to easier drug targeting than Six1 alone. Through this DoD-provided research opportunity, I have developed a strong foundation and passion for translational breast cancer research and wish to continue in an environment where the improvement of patient life is at the forefront of investigation."
Dr. Farabaugh continues her training in breast cancer research as a postdoctoral associate in Dr. Adrian Lee's lab at the Women's Cancer Research Center at the University of Pittsburgh Cancer Institute.
 Lina Dimberg, Ph.D.
Lina Dimberg, Ph.D.
The body has evolved its own mechanisms to keep cancer from initiating by killing tumor cells when they appear. Activating these natural defenses is an ideal way to fight cancer - eradicating tumors without causing negative side effects. One such natural defense mechanism researchers have tried to utilize in the fight against breast cancer is TNF-Related Apoptosis Inducing Ligand, or TRAIL. TRAIL suppresses metastasis by triggering apoptotic cell death in tumor cells. While drugs that mimic TRAIL and activate the TRAIL receptor have shown low toxicity in patients, their therapeutic efficacy has been disappointing, resulting from TRAIL resistance imparted to the tumor over time. After receiving a BCRP Postdoctoral Fellowship Award in fiscal year 2011, Dr. Lina Dimberg performed a genome-wide screen that uncovered several novel mediators that are involved in conferring TRAIL resistance to breast cancers. Dr. Dimberg believes the genes could one day serve as biomarkers to predict therapeutic outcome in clinical trials for TRAIL-based treatments.
"The funding has given me the experience I need to further my career as a breast cancer researcher. At the Era of Hope conference, I could truly feel how scientists and advocates work together towards improving the outcomes of breast cancer patients, and that has been immensely inspiring."
Dr. Dimberg remains a postdoctoral fellow in the lab and continues to explore the causes of TRAIL resistance in breast cancer.
 Chu-An Wang, Ph.D.
Chu-An Wang, Ph.D.
Lymph node metastasis is a major cause of morbidity which also may lead to distant metastasis. The factors that determine if a tumor cell will enter the lymphatic system, however, remain poorly understood. After receiving a BCRP Predoctoral Traineeship Award in fiscal year 2010, Dr. Chu-An Wang discovered a novel mechanism by which tumor cells promote lymphatic growth and metastatic spread. She showed that Six1 promoted the formation of lymphatic vessels that provide a pathway to the lymph node for nearby tumors. She further elucidated Six1's actions by showing that they were dependent on inducing transcription of VEGF-C, a factor known to promote lymphatic vessel growth.
"I gained independent thinking...and had a chance to attend the Era of Hope conference, not only extending my knowledge of breast cancer but also to meet breast cancer patient advocates."
Dr. Wang plans to return to her native country of Taiwan in hopes of obtaining a faculty position and beginning a breast cancer research lab of her own.
 Anna Smith, Ph.D.
Anna Smith, Ph.D.
Small stretches of non-coding RNA, called microRNAs (miRNAs), are known to affect gene expression. Recently they have been implicated in several types of cancer. With the support of a Fiscal Year 2010 BCRP Predoctoral Fellowship Award, Dr. Anna Smith uncovered a collection of miRNAs that are de-regulated in breast cancers, revealing novel ways through which breast cancers metastasize. Dr. Smith showed that not only are these miRNAs regulated by Six1, but many of Six1's oncogenic effects are mediated by the miRNAs. Because of this, Dr. Smith believes that the collection of miRNAs represent new therapeutic possibilities as either novel drug targets or biomarkers to select for patient populations who would benefit from, based on Dr. Micalizzi's work, TGF-ß inhibitors.
"I can't say enough about what this award meant to me and what it did for me. I can truly say that the DoD has motivated me to be a lifelong breast cancer researcher. This mostly came from attending the Era of Hope conference where I was in the room with both researchers and breast cancer advocates. Few programs put these two groups of people together, and I appreciate the advocate presence.... I am so proud to have been a part of this program." Dr. Smith just completed her PhD in the Ford laboratory, and is moving on to a postdoctoral fellowship in Dr. Joaquin Espinosa's laboratory at The University of Colorado, Boulder, where she will continue research in breast cancer, with an emphasis on the p53 tumor suppressive pathway.
Links:
Dr. Doug Micalizzi
Public and Technical Abstracts: The Molecular Mechanism of Six1-Induced Tumor Initiation and Progression
Dr. Erica Koustas
Public and Technical Abstracts: In Vivo Role of Six1 in Mammary Gland Tumorigenesis
Dr. Susan Farabaugh
Public and Technical Abstracts: Six1-Eya Dach Network in Breast Cancer
Dr. Lina Dimberg
Public and Technical Abstracts: Determining the Mechanism of Six1-Induced TRAIL Resistance and Its Role in Breast Cancer Metastasis
Dr. Chu-An Wang
Public and Technical Abstracts: Six1 Regulates VEGF-C, Lymphangiogenesis, and Lymphatic Breast Cancer Metastasis
Dr. Anna Smith
Public and Technical Abstracts: miRNA Involved in Six1-Induced Breast Cancer














