Amyotrophic Lateral Sclerosis
New Nerve-on-a-Chip System Could Revolutionize Therapeutic Development for ALS



May 11, 2023
Dr. J. Lowry Curley, Axosim Technologies

Amyotrophic lateral sclerosis (ALS) is a complex, devastating neurodegenerative disease with a multifactorial pathophysiological presentation. With such a multifaceted etiology, the current preclinical drug screening tools capable of measuring only a few markers at a time are simply insufficient. AxoSim Technologies received a fiscal year 2018 (FY18) ALSRP Therapeutic Idea Award to directly address this problem. The goal of this project was to create a commercial-ready, clinically relevant preclinical model for developing therapeutics for ALS.
The study team, led by Dr. J. Lowry Curley, hypothesized that a three-dimensional (3D) system comprised of neuronal stem cells and specialized brain cells would hold a significantly superior capability of predicting the mechanism of clinical nerve pathology, as compared to conventional systems. To address this hypothesis, the human Nerve-on-a-Chip (NoaC) technology, a 3D micro-physiological system for disease modeling, was developed. The new technology features micro-engineered human motor nerve constructs. These constructs were fabricated using induced pluripotent stem cell (iPSC)-derived coculture organoids consisting of human motor neurons and glial cells. For modeling ALS, the team used iPSC-derived motor neurons and astrocytes that harbor the disease-inducing SOD1 D90A mutation. The objective of this pilot project is to establish the feasibility of obtaining structural and functional metrics that reliably differentiate healthy and ALS human motor nerve tissue in vitro.
Following baseline characterizations and culture combination optimization, the team created NoaC constructs using a variety of ratios of healthy iPSCs-derived motor neurons and healthy iPSC-derived astrocytes. These NoaC constructs were found to be electrically active, which is a sign of axonal maturation. In addition, morphological interactions between the cells appeared more physiologically relevant in the 3D environment of the organoids. Excitingly, the team performed the first known in vitro measurement of population-level electrophysiology of human neuron-astrocyte cocultures, recorded in healthy human motor NoaC construct (Refer to Figure 1).
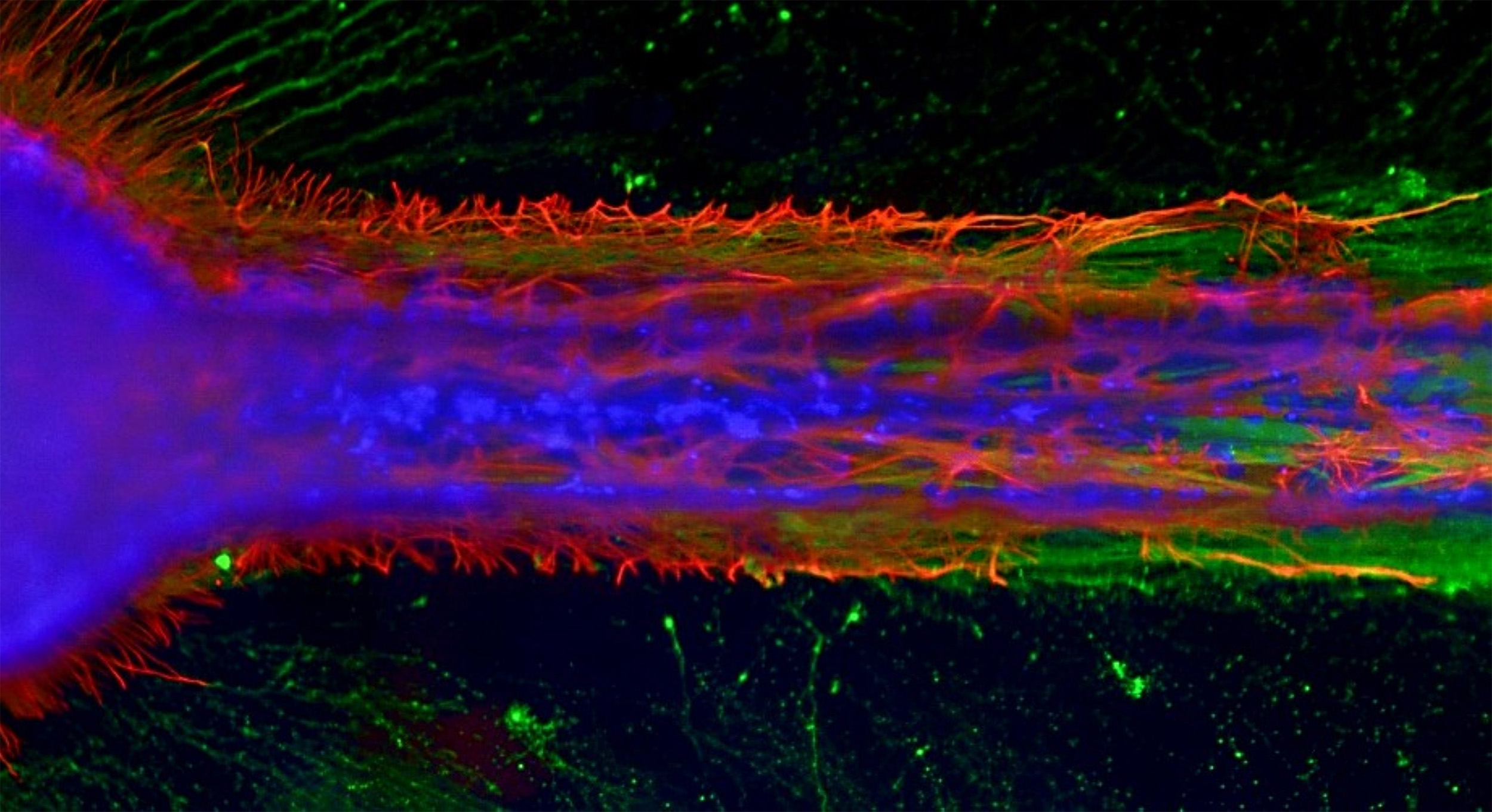
Figure 1: Micro-engineered motor nerve construct. The in vitro mini-nerve fiber tract shows the important developmental process of constrained axonal outgrowth (green) interacting with astrocytes (red) and includes migrating supportive cells (blue). (Figure Provided)
The organoids grown were cultured in NoaC for 42 days, and the team then compared the healthy cultures to the ALS-like cultures. Significant differences were noted in the rates of neurite outgrowth and expression of ALS biomarkers, which could be key metrics when evaluating therapeutics for efficacy in preventing, slowing, or reversing ALS. The team identified SOD1 and PRPH as potential biomarkers within their system, as well as morphological differences between the healthy and ALS human motor neuron models. These metrics can potentially be used to assess the efficacy of therapeutics in the fight against ALS disease progression. This human cell-based preclinical model of ALS will be an important assay to accelerate therapeutic development, ultimately leading to improved health care.
Future plans for the NoaC system include introducing candidate ALS therapeutics in the appropriate combination of healthy and disease motor neuron-astrocyte to demonstrate the rescue of structural and functional pathology.
Regarding the early support from the ALSRP on this project, Dr. Curley writes, "The funding for the development and characterization of an ALS microphysiological system, with clinically relevant pathophysiological biomarkers helped us establish an industry-leading model. Our Nerve-on-a-Chip will serve as a drug-discovery platform that accelerates drug discovery towards clinical trials with an improved chance of success in patients, and we are already in advanced discussions with potential pharmaceutical partners to further validate the system towards adoption into the regular workflow for the development of the next generation of therapeutics for devastating neurodegenerative diseases like ALS."
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
