Amyotrophic Lateral Sclerosis
A New Compound to Prevent Motor Neuron Toxicity in ALS Enters Clinical Trial




June 14, 2022
Brent Stockwell, Ph.D. and Hynek Wichterle, Ph.D., Columbia University
Amyotrophic Lateral Sclerosis (ALS) is often characterized by the pathological accumulation of misfolded proteins in spinal motor neurons, the cells that are selectively vulnerable to the disease. Motor neurons initially engage cellular endoplasmic reticulum (ER) stress pathways to purge misfolded proteins, but if these efforts are unsuccessful, self-sacrificing branches of the ER stress pathway are initiated. This process is thought to contribute to the widespread motor neuron death observed in ALS, and ER stress activation has been observed across many models of ALS and in human patient tissues.


In fiscal year 2015, the ALS Research Program (ALSRP) funded a Therapeutic Idea Award to Dr. Brent Stockwell, professor of biological sciences and chemistry at Columbia University, titled, "Motor Neuron-Protecting Agents as Therapeutics for Treating ALS." Dr. Stockwell and his team, in collaboration with another ALSRP-funded investigator at Columbia University, Dr. Hynek Wichterle, associate professor of pathology & cell biology and neuroscience at Columbia, used the award to develop potent and selective inhibitors of ER stress in a cellular model of ALS and created prosetin, a brain-penetrant kinase inhibitor, as a promising candidate. This initial idea award laid the groundwork for additional development efforts that are continuing along the ALSRP funding pipeline.
In fiscal year 2020, Dr. Stockwell and Dr. Wichterle obtained an ALSRP Therapeutic Development Award to evaluate pharmacology, safety, and efficacy profiles of prosetin, the potent kinase inhibitor that the team found to be efficacious across multiple models of ALS. As part of this new award, Dr. Stockwell and his team will further validate their hypothesis and more confidently determine the appropriate patient population for future clinical applications. Because ER-stress-related pathologies are observed both in familial and sporadic forms of ALS, prosetin's mechanism of action may be broadly applicable to ALS. It should be noted that prosetin is orally bioavailable, metabolically stable, CNS-penetrant, and well-tolerated in long-term administration paradigms, making it a prime candidate for the treatment of ALS.
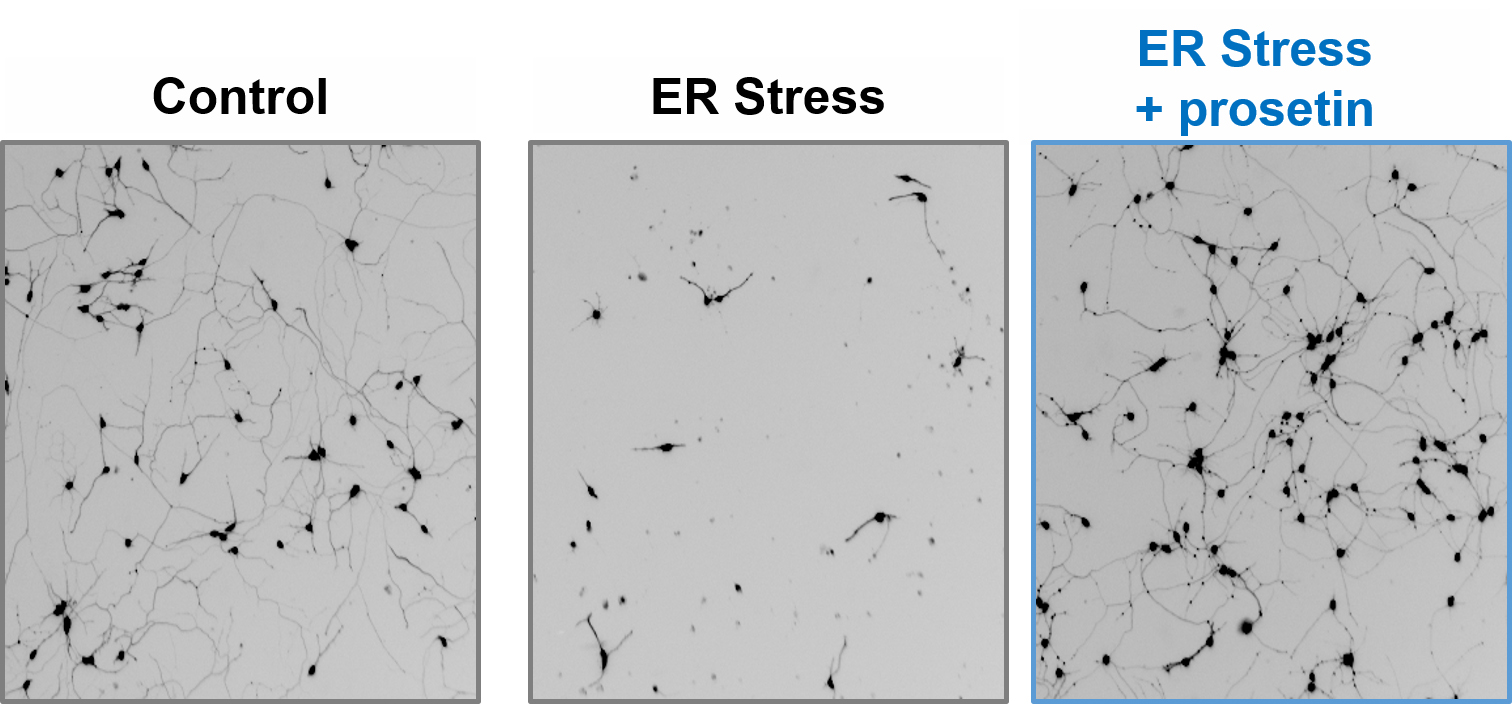
Figure 1: Microscopy images of motor neurons treated with control vehicle alone; Following ER Stress induction; and ER Stress induction in combination with prosetin treatment.
This work is a collaborative effort with Project ALS, a non-profit entity that supports the Project ALS Therapeutics Core at Columbia University (The Core). The Core was established in 2018 and brings together preclinical and clinical ALS research at Columbia University with funding and strategic direction from the non-profit organization. Project ALS pursued orphan drug designation for prosetin, and the U.S. Food and Drug Administration granted the therapeutic orphan-drug status for the treatment of ALS in 2020. This positioned these ALSRP-funded investigators to initiate scientifically rigorous clinical trials of prosetin on an accelerated timescale. Erin Fleming, who was director of external operations for The Core and director of research operations at Project ALS, served as project manager of this effort, coordinating contactor selection and managing workflow amongst our collaborators. Ms. Fleming writes, "The Core aims to bring more rational therapeutic candidates to ALS clinical trials, and as the first investigational drug developed through this collaboration with Columbia, this project was a top priority for Project ALS. ALSRP support has been critical in moving prosetin from concept to clinical trial on an accelerated timescale."
In early 2022, Project ALS partnered with a new startup company ProJenX, which obtained the license to prosetin from Columbia University, to initiate a first-in-human phase 1 clinical trial of this drug in healthy volunteers and ALS patients. This study will begin to determine the safety, tolerability, and pharmacokinetics of single and ascending doses of prosetin versus a placebo in the healthy volunteers. The overall goal of the trial is to determine the optimal dose of prosetin and acquire biomarker data to inform the design of future late-stage trials.
Dr. Stockwell writes, "To our knowledge, our commitment to advance prosetin through early clinical development independently, as a non-profit academic effort, was the first of its kind in ALS. Our ultimate aim is to deliver a meaningful therapy to people with ALS, and because our primary responsibility is to the ALS community, we will conduct this research of prosetin with the urgency and rigor that this disease demands."
References:
Bos P, Lowry ER, Costa J, et al. 2019. Development of MAP4 Kinase Inhibitors as Motor Neuron-Protecting Agents. Cell Chemical Biology 26(12):1703-1715.
Links:
Public and Technical Abstracts: Motor Neuron-Protecting Agents as Therapeutics for Treating ALS
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
