Prostate Cancer
Unique Structural Characterization of Androgen Receptor - A Key Protein in Prostate Cancer



Posted May 18, 2023
Elizabeth V. Wasmuth, Ph.D., Memorial Sloan Kettering Cancer Center

The androgen receptor (AR) is an important transcription factor responsible for development and maintenance of the male phenotype. Hyperactive AR signaling is a hallmark of metastatic prostate cancer, the second leading cause of cancer deaths in American men. Drugs that inhibit AR signaling have been used since the 1940s; however, resistance to even the best therapeutics inevitably arises through rebounds in AR signaling or alternative pathways that render the cancer AR-independent. A major limitation in developing more potent AR inhibitors is that there is not a complete picture of how AR looks or works, knowledge that requires understanding of its atomic details.
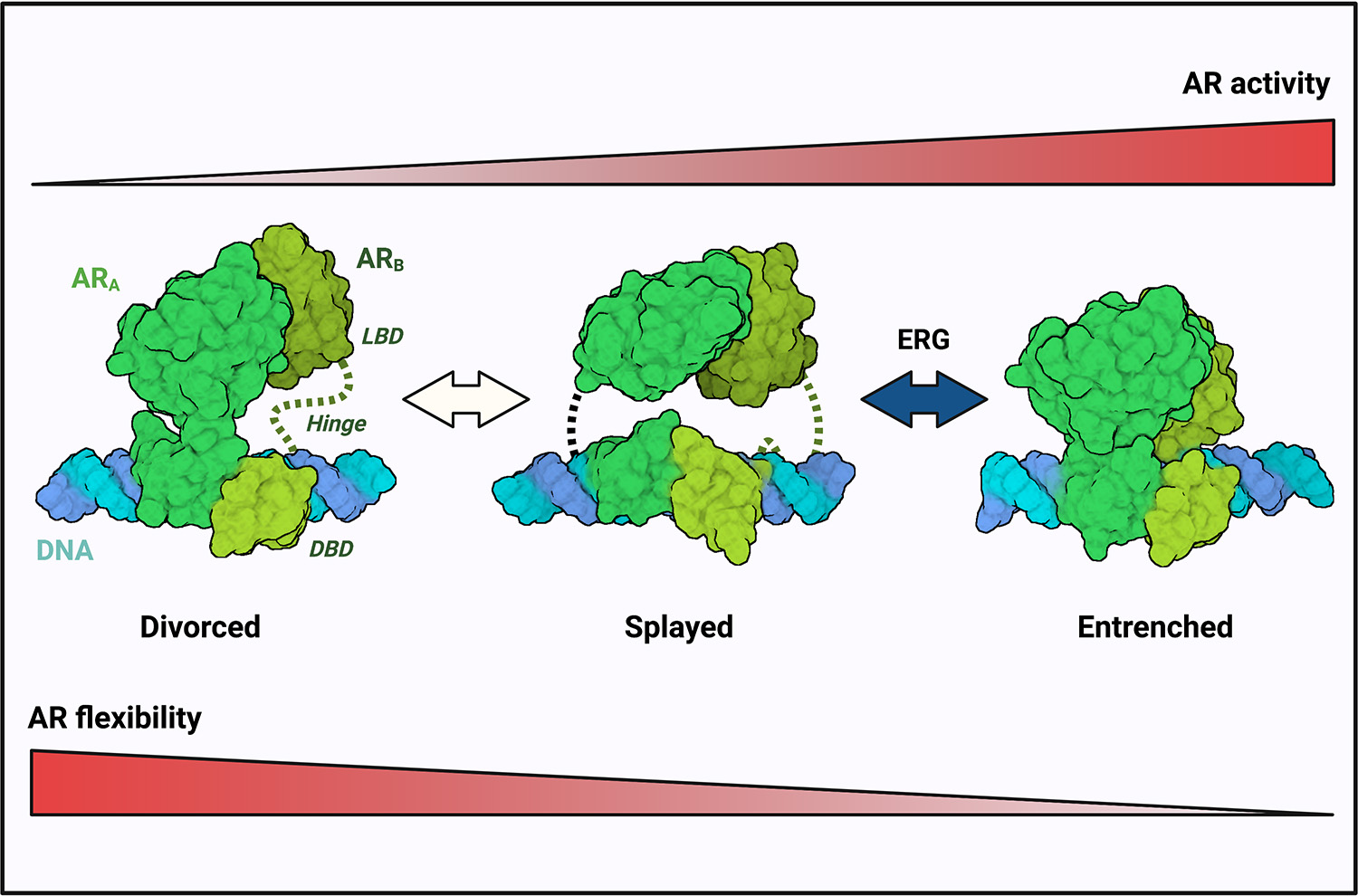
Through work funded by a fiscal year 2017 Prostate Cancer Research Program Early Investigator Research Award, Dr. Elizabeth V. Wasmuth sought to use her expertise in cryo-electron microscopy (cryo-EM) to take a closer look at the AR at the atomic level. Along with her research team at Memorial Sloan Kettering Cancer Center, she devised the first recombinant system by combing full-length AR with a known cancer-causing protein called ERG, which allowed for structural characterization of the AR that had never before been possible. Using cryo-EM, the team has obtained the clearest snapshots of AR to date and discovered that unlike all other related nuclear receptors that bind DNA, including the estrogen receptor, the AR was far more flexible in the shapes that it could form. They believe this feature licenses AR to bind a wider range of DNA substrates and cofactors, enabling it to turn on a wider range of cancer-causing genes.
These results reveal important and previously unknown features of the AR that contribute to its DNA binding and specificity, fundamental knowledge that has influenced collective thinking about AR targeting. These advances led to two publications in prestigious journals (Wasmuth et al, PNAS, 2020; Wasmuth et al, Molecular Cell, 2022) and have opened new avenues of research that will allow them to answer outstanding and fundamental questions pertaining to AR regulation. Importantly, this system they developed will facilitate discovery of novel therapeutics that can more specifically and effectively target the AR.
Publications:
Elizabeth V. Wasmuth,, Arnaud Vanden Broeck,, Justin R. LaClair, Susan T. Weintraub, Sebastian Klinge, Charles L. Sawyers; Allosteric interactions prime androgen receptor dimerization and activation; 2022, Molecular Cell 82, 1-11
Wasmuth EV, Hoover EA, Antar A, Klinge S, Chen Y, Sawyers CL. Modulation of androgen receptor DNA binding activity through direct interaction with the ETS transcription factor ERG. Proc Natl Acad Sci U S A. 2020 Apr 14;117(15):8584-8592. doi: 10.1073/pnas.1922159117. Epub 2020 Mar 27. PMID: 32220959; PMCID: PMC7165421.
Links:
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
