Posted May 20, 2014
Bryan Shaw, Ph.D., Baylor University, Waco, Texas
Scientific evidence is mounting to show that the progressive neurodegeneration of ALS is associated with the incorrect folding of proteins or malfunctions in the disposal of misfolded proteins within nerves. Cells have specific mechanisms for recognizing and disposing of misfolded proteins, and malfunctions in this disposal process lie at the heart of the forms of ALS. The hereditary form of ALS (familial, or fALS) involves mutations in superoxide dismutase-1 (SOD1) that lead to its misfolding. Why misfolding of this particular protein causes ALS is not entirely clear, but there is evidence that aggregation of misfolded proteins accompanies nerve damage.
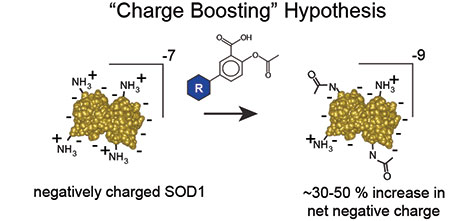
Using an FY10 ALSRP Therapeutic Idea Award, Dr. Bryan Shaw has been exploring his hypothesis that if the aggregates of misfolded proteins could be prevented from forming, neurodegeneration would be stalled long enough to allow the cells to dispose of misfolded proteins properly. Most proteins, including SOD1, have many positive and negative charges on their surfaces, but possess a net negative charge. Dr. Shaw reasoned that if the net charge of SOD1 proteins could be increased, the tendency to aggregate would decrease because like-charged protein molecules repel each other. Dr. Shaw refers to this effect as "charge boosting" and the phenomenon has been observed in other naturally occurring proteins. Knowing that normal SOD1 proteins bear a net negative charge, Dr. Shaw acetylated them, thereby neutralizing the positive charges of lysine sidechains on their surface, rendering the proteins more negative. He found that neutralizing even two lysines in the SOD1 A4V mutant (known to be associated with ALS) significantly reduced aggregation.
For this finding to develop into a practical therapy, a drug based on this principle needs to specifically acetylate SOD1 and avoid the myriad of other proteins bearing surface lysine residues. Therefore, Dr. Shaw's group is designing molecules for this purpose using computer modeling programs to design easily synthesizable molecules that should bind exclusively to SOD1. The next steps will be to synthesize then test these molecules to identify those that will optimally prevent SOD1 aggregation.