Traumatic Brain Injury and Psychological Health
Developing the Use of Intranasal Insulin for On-Field Treatment of Traumatic Brain Injury



Posted March 22, 2023
Kimberly Byrnes, Ph.D., Uniformed Services University

(Photo Provided)
Traumatic brain injuries (TBI) have accounted for a large proportion, approximately 22%, of injuries sustained in the most recent military conflicts in Afghanistan and Iraq. To date, more than 486,000 Service Members were diagnosed with a TBI between 2000-2022, as determined by the Department of Defense TBI Center of Excellence. TBIs cause a host of long-term, debilitating symptoms including problems with memory, thinking, balance, and behavior. This significantly impacts a Service Member’s ability to return to duty and readiness, as well as their overall quality of life and well-being. As with most traumatic injuries, the sooner they are treated, the better the effect on long-term outcomes. However, options for easy-to-administer treatments at the location of the injury are limited. There is an unmet need for a stable, portable, and immediately effective treatment for TBIs, both on the battlefield and in the civilian world.
With a fiscal year 2021 award funded by the Traumatic Brain Injury and Psychological Health Research Program (TBIPHRP), Dr. Byrnes and her research team hope to address this need for TBI point-of-injury treatments with the use of an intranasal insulin (INI). This drug is currently in clinical trials for use in other indications, with good safety and tolerability in humans.
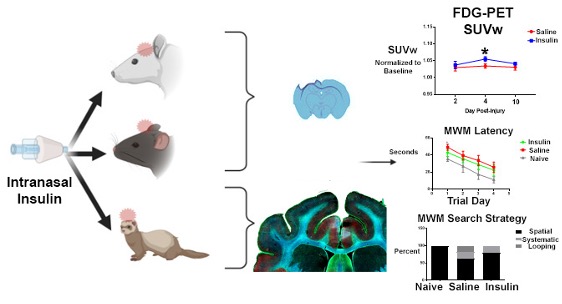
The aim of this study is to determine the best INI dosing paradigm for use in TBI. To do this,
The team will reproduce both blast-related and rotational head injury models of TBI in adult mice, rats, and ferrets before dosing with INI or a placebo (Figure 1). Following treatment, the team will assess the animals’ outcomes, including their behavior and brain injury pathology. By using varied animal models of TBI and different TBI severity levels, the team will be able to understand which doses of INI are most effective. The inclusion of ferrets will accelerate the timeline to using INI in humans with TBI, because their brain structure more closely approximates the human brain due to the many deep folds and ridges (known as gyrencephalic). Importantly, this project also uses female animals, allowing the team to parse out any potential sex-related difference in the effectiveness of INI administration.
This study will address the unmet need for point-of-care treatments for TBI and will be the first to study INI administration in highly translatable TBI models. This information is critical to moving us closer to effective treatments that can help stave off long-term problems associated with TBI. Ultimately, the study team plans to develop a hand-held portable kit for use at the time of injury, to be carried by active-duty Service Members and civilian medics.
References:
Shaughness M, Acs D, Brabazon F, Hockenbury N, Byrnes KR. Role of Insulin in Neurotrauma and Neurodegeneration: A Review. Front Neurosci. 2020 Sep 23;14:547175.
Brabazon F, Wilson CM, Jaiswal S, Reed J, Frey WH Nd, Byrnes KR. Intranasal insulin treatment of an experimental model of moderate traumatic brain injury. J Cereb Blood Flow Metab. 2017 Sep;37(9):3203-3218. doi: 10.1177/0271678X16685106. Epub 2017 Jan 6. PMID: 28058996; PMCID: PMC5584695.
Links:
Last updated Wednesday, March 22, 2023

















