Prostate Cancer
An International Registry to Inform Treatment Choices for Patients with Advanced Prostate Cancer



Posted June 30, 2023
Daniel George, M.D. - Duke Cancer Institute
Lorelei Mucci, Sc.D. - Harvard T. H. Chan School of Public Health
Philip Kantoff, M.D. - Convergent Therapeutics

(Photo provided)

(Photo provided)

(Photo provided)
Advances in treatments and early detection have contributed to a 5-year survival rate of over 99% for individuals diagnosed with localized or regional prostate cancer.1 However, patients living with advanced prostate cancer or who encounter disease recurrence face a lower survival rate, leading them to pursue additional treatments with corresponding side effects, impacting their quality of life. Best estimates report 180,000 patients living in the United States with advanced prostate cancer. In recent years, new U.S. Food and Drug Administration (FDA)-approved therapies offer survival advantages for men with advanced prostate cancer, but survival rates do not fully describe the needs of this patient population; management of cancer and the impact of side effects on their quality of life must also be considered. Thus, better understanding of current patient management models will help improve survival and patient outcomes for this population with a great need for improved treatments and disease management options.
The Prostate Cancer Research Program (PCRP) has invested in a diverse array of research projects to address the needs of advanced prostate cancer patients. More specifically, the PCRP has financially supported the collaborative efforts of leading clinical research centers through its multi-institutional Prostate Cancer Clinical Trials Consortium (PCCTC) since fiscal year 2005. This consortium was originally tasked with accelerating the early-phase testing of novel prostate cancer therapies with the goal of bringing needed treatments to patients at a faster pace than would be possible outside the consortium. The PCCTC's collaborative clinical trial design and execution efforts have been remarkably successful, resulting in FDA approval of seven prostate cancer treatments that are now available and utilized by cancer patients in the Military Health System and the general American public.
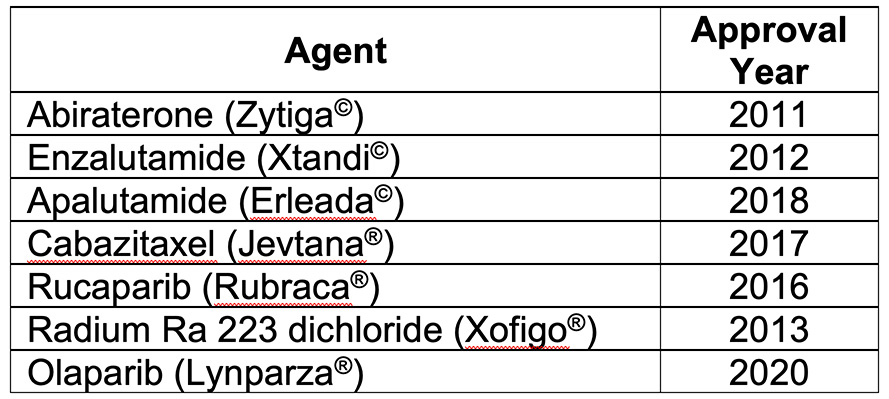
The PCCTC network has also enabled additional collaborations that impact clinical trial design for prostate cancer. Earlier in the history of the PCCTC, the investigators worked together to develop consensus guidelines that included revisions of disease state models, eligibility criteria, outcome criteria, and reporting methods. These guidelines accelerated the movement of novel agents through clinical trials to deliver promising therapeutics to patients more quickly. The PCCTC investigators have not stopped there. In 2017, they launched their biggest collaborative effort yet: the International Registry for Men with Advanced Prostate Cancer (IRONMAN). The PCCTC strategically leveraged its PCRP-funded infrastructure and financial support from Movember and many industry partners to initiate the monumental effort of collecting patient data from 5,000 patients newly diagnosed with advanced prostate cancer, defined as metastatic hormone sensitive and castration resistant prostate cancer, from 16 different countries. Their ultimate goal is to identify optimal treatment sequences to improve survival, understand patient-reported outcomes, and explore novel biomarkers to understand treatment-resistance mechanisms. The registry is led by Principal Investigators Professor Lorelei Mucci of the Harvard Chan School of Public Health, Dr. Daniel George of Duke Cancer Institute, and Dr. Philip Kantoff, the former Chairman of Medicine at Memorial Sloan Kettering Cancer Center and Emeritus Professor at Harvard Medical School, together with Jake Vinson (PCCTC) and Paul Villanti (Movember) on the Executive Committee.
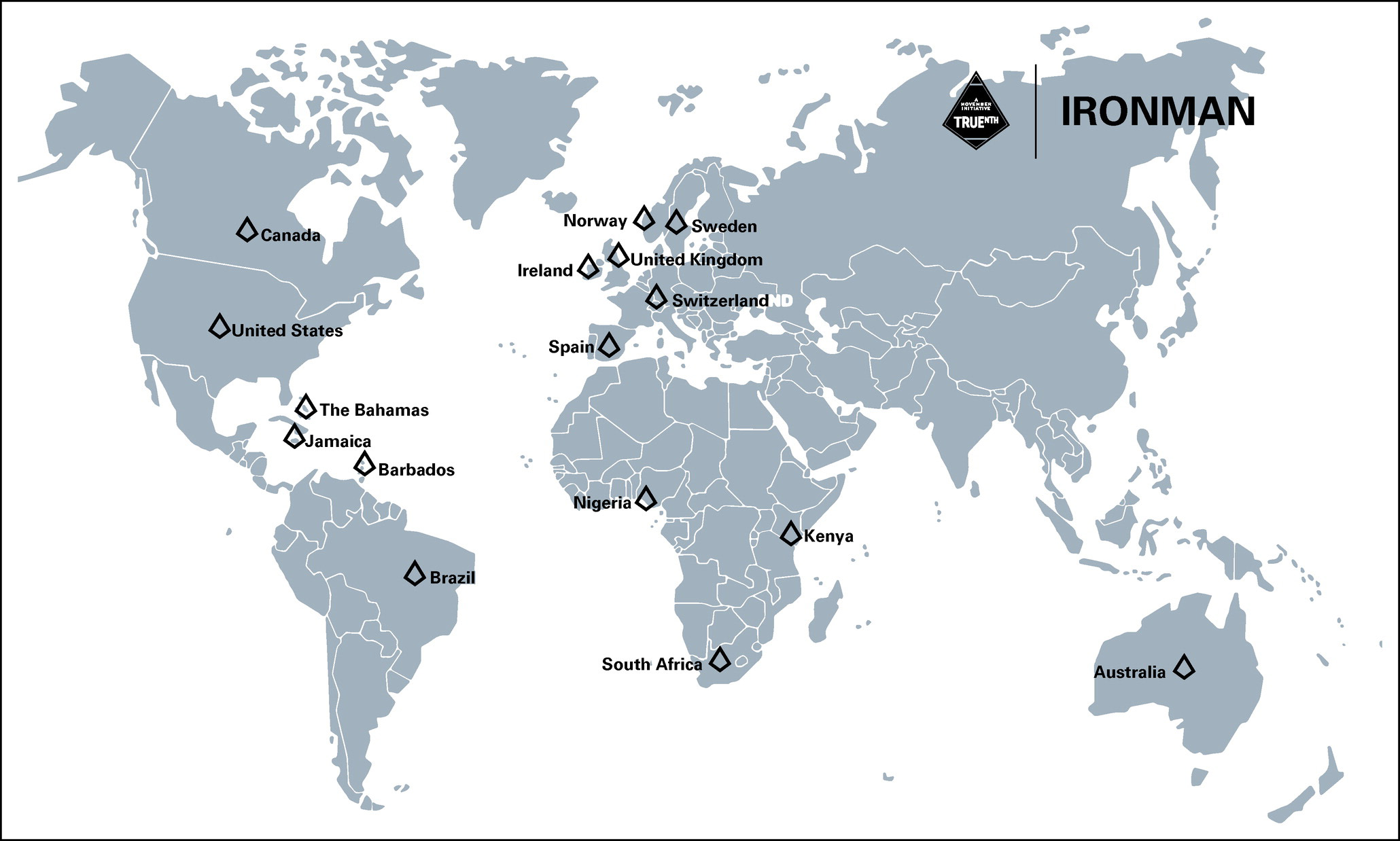
In July 2022, the IRONMAN team published their progress to date, reporting the enrollment of the first 2,682 individuals in 12 of the 16 participating countries. Sites were strategically selected to ensure representation from patient populations that are disproportionately affected by prostate cancer, including Veterans from Veterans Affairs Medical Centers and men of African descent in Australia, The Bahamas, Barbados, Brazil, Canada, Ireland, Jamaica, Kenya, Nigeria, Norway, Spain, Switzerland, Sweden, South Africa, the United Kingdom, and the United States. Patients participating in the registry are contributing biospecimen samples, medical records, and testing results, as well as patient questionnaires designed to gather outcome data including pain impact, urinary function, cognitive health, and sleep quality. Recognizing that physician approaches are key to unravelling patient outcomes and decision-making, IRONMAN investigators are also administering questionnaires to physicians to address gaps in knowledge regarding reasons for selecting and changing treatments.
While a study of this magnitude faces challenges such as new regulations, the COVID-19 pandemic, and global differences in research standards, IRONMAN's collaboration with the PCRP-funded PCCTC has positioned this effort to succeed. The IRONMAN team was recently awarded a 2022 Prostate Cancer Foundation Challenge Award to support analysis of the clinical and patient-reported registry data, demonstrating the power of funding contributions from multiple agencies with the shared goal of conquering prostate cancer. Insights from IRONMAN will have the potential to influence practice patterns to improve survival and outcomes in patients with advanced prostate cancer, especially in communities and groups of people who may not be benefiting from recent advances in therapeutic options.
For more information on study status and participating study locations, please refer to ClinicalTrials.gov (NCT03151629). Individuals interested in collaborating on research projects using IRONMAN should contact (pcctcironmanregistry@mskcc.org).
References:
1American Cancer Society. 2023. Cancer Facts & Figures 2023. Atlanta, Ga. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/2023-cancer-facts-figures.html
Publications:
Mucci LA, Vinson J, Gerke TA, et al. 2023. First Look at the Baseline Characteristics of Participants in IRONMAN, the International Registry for Men with Advanced Prostate Cancer. Journal of Clinical Oncology, 41(6):85.
Mucci LA, Vinson J, Gold T, et al. 2022. IRONMAN: A Novel International Registry of Men with Advanced Prostate Cancer. JCO Global Oncology, 8:e2200154. Published online 2022 Nov 4. doi: 10.1200/GO.22.00154.
Rencsok E, George DJ, Kantoff PW, et al. 2022. First Look at Patient-Reported Outcomes from IRONMAN, the International Registry of Men with Advanced Prostate Cancer. Journal of Clinical Oncology, 40(6):69.
McKay RR, Gold T, Zarif JC, et al. 2021. Tackling Diversity in Prostate Cancer Clinical Trials: A Report from the Diversity Working Group of the IRONMAN Registry. JCO Global Oncology, 7:GO.20.00571. Published online 2021 Apr 9. doi: 10.1200/GO.20.00571.
Scher HI, Morris MJ, Stadler WM, et al. 2016. Trial Design and Objectives for Castration-Resistant Prostate Cancer: Updated Recommendations from the Prostate Cancer Clinical Trials Working Group 3. Journal of Clinical Oncology, 34(12):1402-18.
Links:
The Prostate Clinical Trials Consortium
Last updated Friday, December 12, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
