Tuberous Sclerosis Complex
Gene Therapy, a New Approach to Treating TSC
Posted March 1, 2019
Xandra O. Breakefield, Ph.D., Massachusetts General Hospital

Credit: Pike See Cheah
Tuberous sclerosis complex (TSC) is a rare genetic disorder marked by loss of function of hamartin or tuberin - proteins encoded by the TSC1 and TSC2 genes, respectively. Mutations in these genes lead to development of non-malignant tumors throughout the body; however, the clinical manifestations that most strongly impact quality of life are brain seizures, developmental delays, autism, and intellectual disabilities. Current drug treatments, such as rapamycin analogues, are effective but have a number of limitations including expense, the need for continual treatment, reduced access to the brain, and potential interference with brain development in young children. Gene therapy is an attractive alternative to conventional TSC treatments in that it can be given with only a single application, has limited side effects, and has been proven effective in several genetic diseases. Dr. Xandra Breakefield and her collaborators have been working on gene therapy for neurologic diseases for some time. Aware that treating young TSC-afflicted children with drugs might affect brain development and can have other side effects, she felt that gene therapy to replace the missing gene in TSC with the equivalent of a normal copy would not disturb brain development and could be effective with a single injection, rather than continuous treatment with drugs.
With funding from Fiscal Year 2012 and Fiscal Year 2015 Tuberous Sclerosis Complex Research Program Exploration – Hypothesis Development Awards, Dr. Breakefield and her team are investigating gene therapy as a potential therapeutic approach for TSC. Dr. Breakefield and her team have developed a preclinical mouse model of TSC in which the TSC1 or TSC2 gene are missing in some brain cells starting at the time of birth. These mouse models led to early death due to accumulation of fluid in the brain (hydrocephalus) and abnormal electrical activity (epilepsy). By introducing the missing gene into the bloodstream via an adeno-associated virus (AAV) vector, a carrier used to deliver missing genes to recipient cells, prior to death, Dr.Breakefield was able to normalize brain structures and extend the lifespan to almost normal length in mouse models of both TSC1 and TSC2.
“It takes a village,” states Dr. Breakefield. “This work would not have been possible without the dedicated efforts of Shilpa Prabhakar, M.S., who has been moving it forward since 2004 as the focus of her career. We have also been blessed with truly outstanding colleagues who have joined the effort – Dr. Casey A. Maguire, an expert in AAV vectors, and Dr. Pike See Cheah, with excellent skills in all aspects of the work. Of course, we are deeply appreciative of the DoD funding without which our research on tuberous sclerosis would not have been possible.”
Having effective, clinically available treatments is of utmost importance to TSC patients. To that end, Dr. Breakefield and her lab are currently optimizing a vector for whole body systemic delivery, as well as investigating the biological impact of gene replacement. With a patent currently submitted for her work, Dr. Breakefield is looking to partner with a biotechnology company in hopes of developing a clinical grade vector that can be evaluated in clinical trials of TSC. Moreover, she is hoping to work with the National Institutes of Health to evaluate next steps in preclinical studies to optimize this therapeutic strategy via the IGNITE program.
AAV vectors have proven safe and beneficial in gene therapy for a number of human diseases, and U.S. Food and Drug Administration-approved products are appearing this year, so Dr. Breakefield’s work gives promise to the possibility that application of this approach could lead to its effective use in treating TSC.
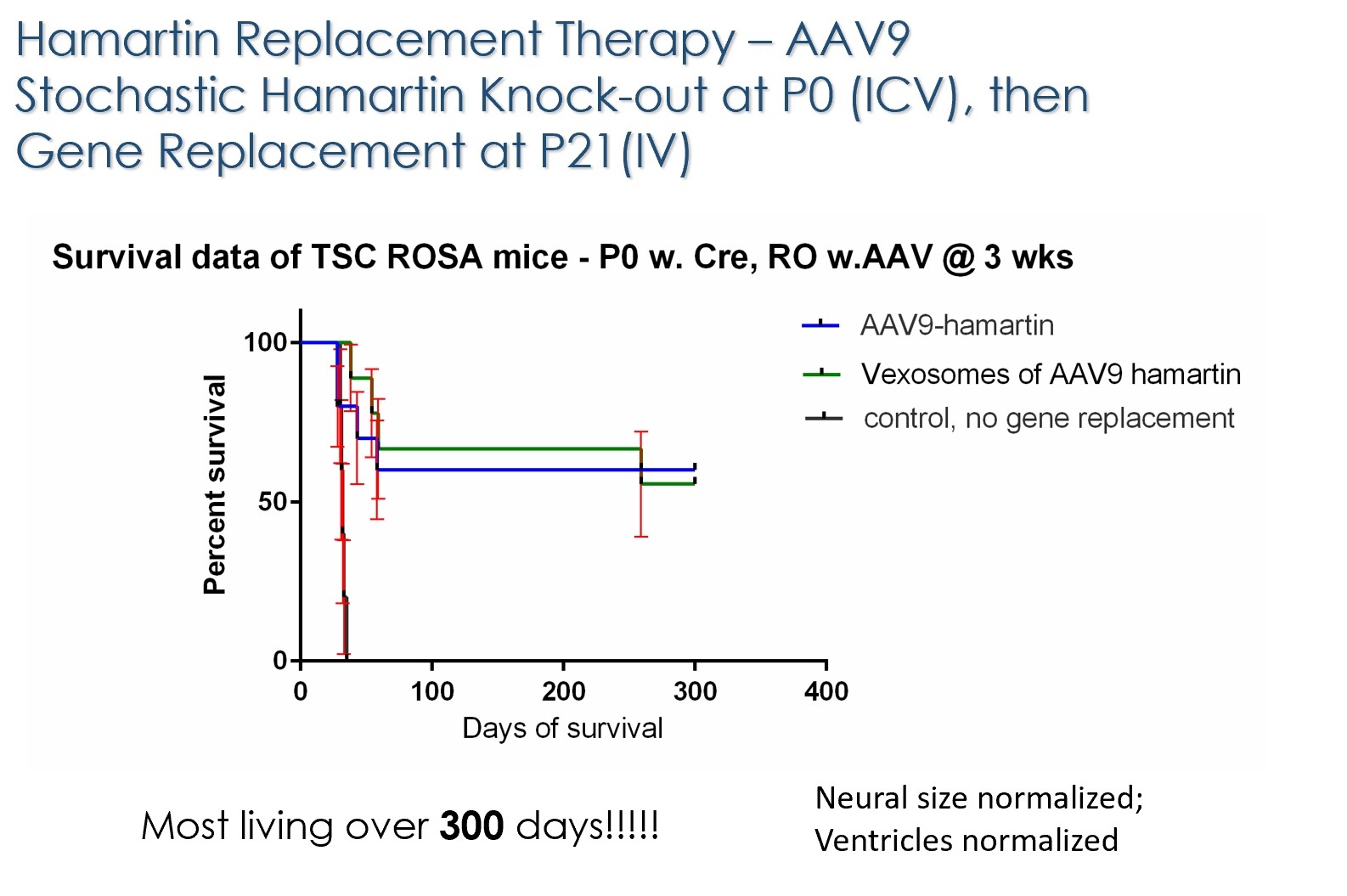
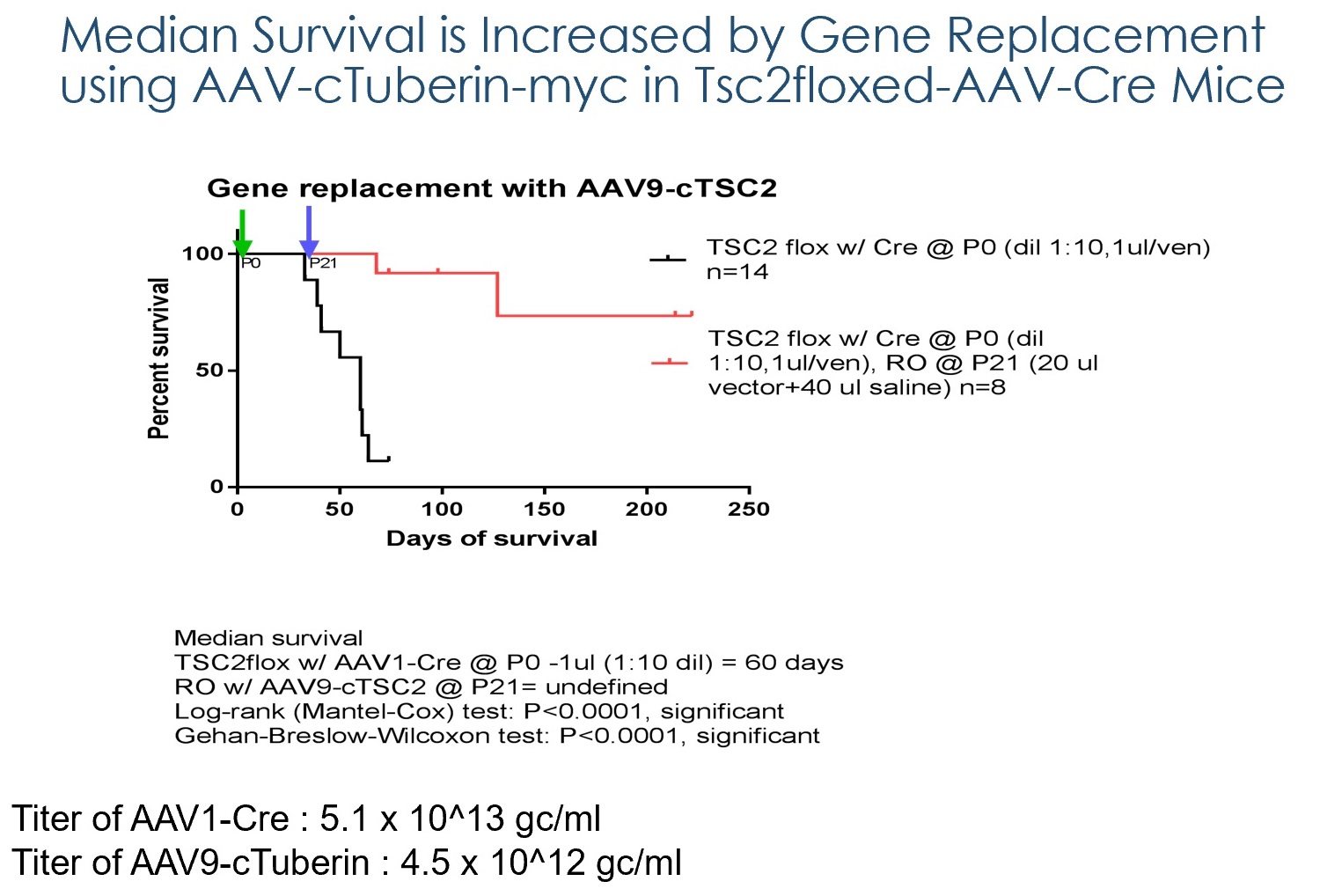
Publications:
1) Prabhakar S, Goto J, Zhang X, Sena-Esteves M, Bronson R, Brockmann J, Gianni D, Wojtkiewicz GR, Chen JW, Stemmer-Rachamimov A, Kwiatkowski DJ, and Breakefield XO (2013). Stochastic model of Tsc1 lesions in mouse brain. PLoS One 8(5):e64224.
2) Prabhakar S, Zhang X, Goto J, Han S, Lai C, Bronson R, Sena-Esteves M, Ramesh V, Stemmer-Rachamimov A, Kwiatkowski DJ, and Breakefield XO (2015). Survival benefit and phenotypic improvement by hamartin gene therapy in a tuberous sclerosis mouse brain model. Neurobiology of Disease 82:22-31.
Links:
Public and Technical Abstracts: Gene Therapy to Extend Lifespan of Tsc1 Conditional Brain Knockouts
Public and Technical Abstracts: Systemic Gene Therapy for Tuberous Sclerosis
Last updated Thursday, May 26, 2022

















